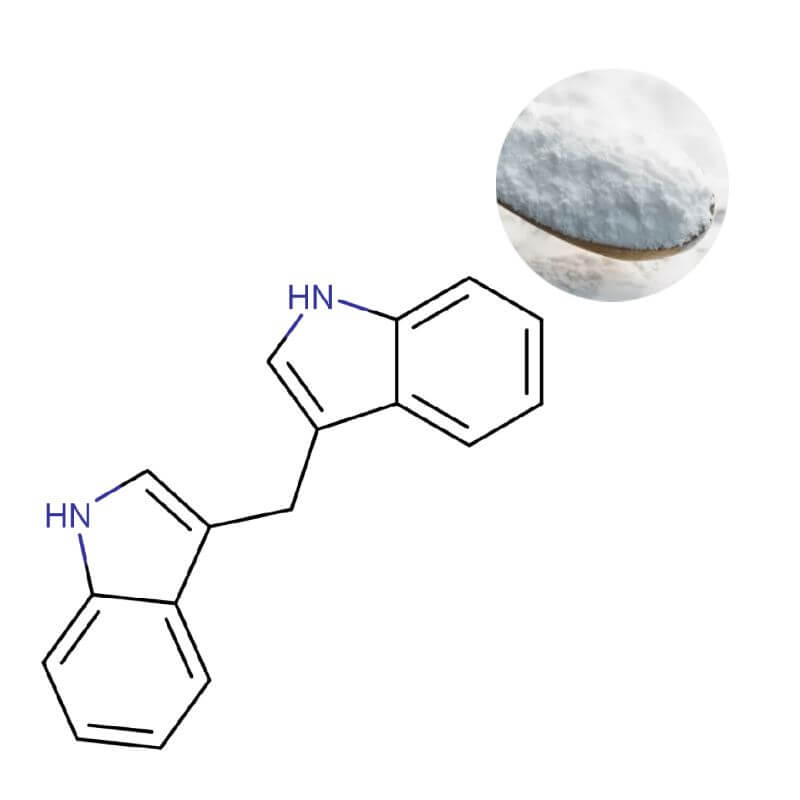
DIM Diindolylmethane Powder
| Product Name | DIM (3,3'-Diindolylmethane) Powder |
| CAS Number | 1968-05-4 |
| Appearance | White to off-white crystalline powder |
| Purity | ≥99% (by HPLC) |
| Packaging | 1 kg/foil bag; 25 kg/paper drum |
| MOQ | 1 kg |
DIM (3,3'-Diindolylmethane) Powder: Enabling Clinically‑Supported Hormonal Balance Formulations
For brands aiming to differentiate in the women's health and sports nutrition segments, sourcing a reliable active ingredient that delivers on its scientific promise is critical. DIM (3,3'-Diindolylmethane) has emerged as a leading compound for supporting healthy estrogen metabolism, yet raw material quality and formulation expertise remain the true differentiators. This article provides an objective roadmap—from the latest human evidence to practical formulation and market positioning—so that R&D and procurement leaders can confidently integrate high‑purity (≥99%, HPLC‑verified) DIM powder into their product pipelines. With regulatory scrutiny intensifying and consumers demanding clinically substantiated ingredients, a well‑sourced DIM is becoming a cornerstone for credible hormone health portfolios.
Clinical Foundation: How DIM Modulates Estrogen Metabolism Across Life Stages
The primary mechanism of DIM lies in its ability to shift estrogen metabolism toward the production of 2‑hydroxyestrone (2‑OHE1) while down‑regulating the 16α‑hydroxyestrone (16α‑OHE1) pathway. This balance is widely recognized as a biomarker for healthy estrogen clearance. Two recent human studies provide compelling, population‑specific evidence:
- A retrospective cohort study involving 1,458 postmenopausal women using transdermal estradiol demonstrated that concomitant DIM intake significantly altered six urinary estrogen metabolites and increased the 2‑OHE1/16α‑OHE1 ratio (P < 0.001) (doi: 10.1097/GME.0000000000002542). This confirms DIM's efficacy even when exogenous hormones are present.
- In a larger cohort of 909 premenopausal women, DIM supplementation again produced broad shifts in the urinary estrogen profile, with a marked increase in the protective 2‑OHE1:16α‑OHE1 ratio (doi: 10.1186/s12906-024-04708-7).
Practically speaking, this means formulators can now target distinct life stages—perimenopause, menopause, or general hormonal health—with the same core ingredient, backed by real‑world evidence. For R&D teams, these studies provide the clinical ammunition needed to substantiate claims across multiple life‑stage products, reducing the need for costly repeat trials. Beyond estrogen modulation, DIM exhibits antioxidant and cell‑protective properties (e.g., activating ATM‑dependent DNA repair pathways), which further supports its use in holistic wellness formulas. For the B2B buyer, selecting a DIM supplier that can provide batch‑specific CoA documentation linked to such clinical benchmarks is the first step toward creating a credible, compliant finished product.
Quality Benchmarks: What Defines a Premium DIM Raw Material
With a mature ingredient like DIM, purity and physical characteristics directly influence both regulatory acceptance and formulation success. The industry standard for a premium grade is ≥99% assay by HPLC, accompanied by strict control of residual solvents (USP <467>) and heavy metals (Pb ≤1.0, inorganic As ≤1.0, Cd ≤0.5, Hg ≤0.1 mg/kg). However, experienced sourcing professionals look beyond the certificate of analysis to three practical parameters:
| Parameter | Typical Specification | Why It Matters for Your Product |
|---|---|---|
| Particle size | ≥95% through 80 mesh | Ensures uniform flow during direct compression or encapsulation, minimizing weight variation. |
| Loss on drying | ≤0.5% | Low moisture content prevents microbial growth and extends shelf life stability. |
| Melting point | 166.0–170.0°C | A narrow, sharp range confirms high purity and absence of polymorphs that could affect dissolution. |
From a compliance standpoint, a GMP‑certified supply chain that delivers a full, batch‑specific CoA is non‑negotiable. It not only satisfies FDA and international regulatory expectations but also reduces the risk of finished product rejection during quality audits. Key sourcing considerations include requesting stability data under ICH conditions and verifying that the manufacturer uses validated HPLC methods. For brands aiming for “clean label” positioning, a synthetic origin (free of common allergens and pesticides) is often preferable—a detail that can be highlighted in marketing without the risk of botanical contaminant variability. These specifications are not merely technical details; they serve as key performance indicators (KPIs) that predict manufacturing efficiency and end‑product quality—critical metrics for procurement teams evaluating total cost of ownership.
Formulation in Practice: Overcoming Bioavailability and Stability Hurdles
DIM is a lipophilic molecule with inherently low aqueous solubility, making bioavailability the central formulation challenge. An effective formulation strategy requires more than simply encapsulating the raw powder. Based on current industry practice and pharmacokinetic studies, three approaches are widely adopted:
- Lipid‑based delivery: Incorporating DIM into softgels with a carrier oil (e.g., MCT oil) and a solubilizer (e.g., polysorbate 80) can increase absorption by avoiding the dissolution rate‑limiting step in the GI tract.
- Micronization: Reducing particle size to the micron range enhances surface area and can modestly improve dissolution, though it is often combined with surfactants for meaningful gains.
- Formulation with absorption enhancers: Proprietary complexes (such as those using cyclodextrins or phytosomes) have been shown in animal models to boost bioavailability by up to 50‑fold compared to crystalline DIM.
Typical daily doses in clinical studies range from 100 to 300 mg, but the optimal dose in a finished product depends on the chosen delivery system. It is worth noting that DIM is stable under normal processing conditions (temperatures below 80°C) but should be protected from prolonged exposure to high humidity during blending. From a commercial perspective, investing in a bioavailability‑enhanced format—even at a slightly higher raw material cost—can justify a premium finished product price and improve consumer adherence through lower pill burden. An effective formulation requires balancing these technical choices with target market positioning. Each delivery approach carries distinct cost‑structure and scalability implications, making the choice a strategic decision that aligns with brand positioning and target price point.
Market Opportunities: Positioning DIM in Women's Health and Beyond
Consumer awareness of hormone health has expanded beyond menopause into areas such as PMS support, acne management, and even male prostate health. DIM’s dual role in estrogen balance and antioxidant support makes it a versatile platform for product innovation. Consider the following white‑space opportunities:
- Perimenopause support complexes: Combine DIM with botanicals like black cohosh or chasteberry, and include a probiotic targeting the estrobolome to create a comprehensive hormonal aging solution.
- Sports nutrition & recovery: DIM can be positioned in formulas aimed at athletes seeking natural hormone optimization (e.g., with zinc, magnesium, and ashwagandha) to support healthy testosterone/estrogen ratios.
- Acne & skin health: Given the link between hormonal fluctuations and sebum production, DIM is gaining traction in beauty‑from‑within supplements targeting adult acne.
For B2B buyers, the ability to offer a finished product that stands out in a crowded category often hinges on the quality of the DIM source and the accompanying technical dossier. Brands that work with a supplier capable of providing formulation support (custom blends, stability testing, and regulatory guidance) can accelerate time‑to‑market while minimizing development risks. By leveraging DIM’s versatility, brands can enter adjacent categories with minimal R&D duplication, maximizing return on ingredient investment. Ultimately, the most successful products will be those that translate the strong human evidence for DIM into a clear, consumer‑friendly narrative—backed by a reliable, certified raw material.
Next Steps for Your Product Development
Choosing a premium DIM ingredient with ≥99% purity, clinical validation, and a robust technical package directly reduces formulation risk and strengthens your finished product’s market position. To evaluate how this raw material performs in your specific application, we invite you to request a complimentary sample and comprehensive technical dossier. This package includes a batch‑specific CoA, stability summary, and formulation guidelines—everything needed for cross‑functional teams (R&D, quality, marketing) to assess alignment with brand goals and move from concept to commercial launch with confidence. Request your complimentary sample & technical dossier and take the next step toward a differentiated, science‑backed supplement.
Frequently Asked Questions
Share this product
Related Products
Ready to get started?
Contact our team for technical specifications, pricing, and customized solutions.