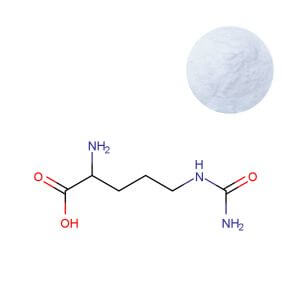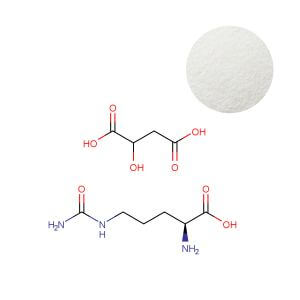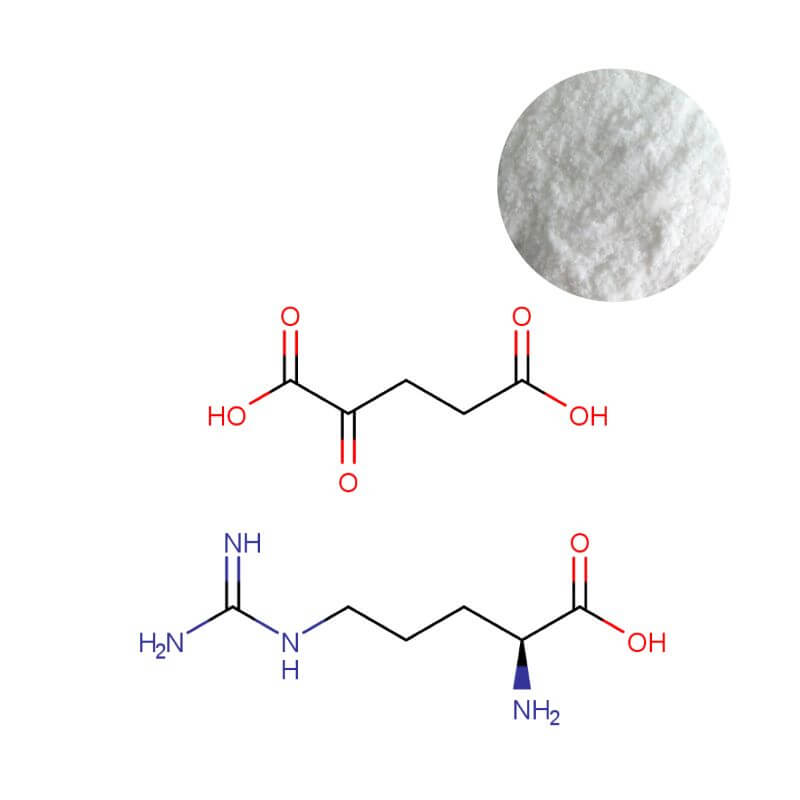
AAKG Powder
| Product Name | Arginine Alpha-Ketoglutarate (AAKG) 2:1 Powder |
| CAS Number | 5256-76-8 |
| Appearance | White to off-white crystalline powder |
| Purity | ≥ 98.0% (by HPLC) |
| Packaging | 1 kg/foil bag, 25 kg/paper drum |
| MOQ | 1 kg |
Arginine Alpha‑Ketoglutarate 2:1 Powder: Enabling Differentiated Sports Nutrition Formulations
For brand owners and R&D teams, the challenge is no longer just adding arginine to a pre‑workout blend. The market demands clean labels, reproducible efficacy, and batch‑to‑batch consistency. That is why the 2:1 molar ratio of L‑arginine to alpha‑ketoglutarate (AAKG 2:1, CAS 5256‑76‑8) has become the preferred specification among formulators of premium sports supplements. With a verified purity ≥98.0% by HPLC and a near‑neutral pH (5.5–7.0), this crystalline powder offers predictable solubility and compatibility. When you source bulk AAKG 2:1 from a GMP‑certified partner, you secure a traceable supply chain backed by a batch‑specific Certificate of Analysis (COA) – the foundation for reliable product launches. For brands competing in the crowded pre‑workout segment, adopting a fully documented 2:1 ratio ingredient eliminates formulation guesswork and creates a defensible scientific positioning against generic alternatives.
Mechanism of Action: Why AAKG 2:1 Supports Vascular & Muscle Performance
AAKG is an amino acid derivative that delivers L‑arginine in a stabilised salt form. Once absorbed, L‑arginine serves as the obligate substrate for endothelial nitric oxide synthase (eNOS), producing nitric oxide (NO) – a key vasodilator. Enhanced NO‑mediated vasodilation increases blood flow, oxygen, and nutrient delivery to working muscles, which is perceived as the “muscle pump” effect in pre‑workout products. Beyond haemodynamics, the alpha‑ketoglutarate (AKG) moiety enters the Krebs cycle and plays a central role in nitrogen metabolism, helping to reduce exercise‑induced ammonia accumulation. A 2024 narrative review systematically evaluated 112 peer‑reviewed papers and highlighted that AKG supports muscle regeneration by activating muscle satellite cells and modulating macrophage polarisation, while also inhibiting protein degradation pathways (doi: 10.3390/nu16223968). From a strategic formulation perspective, this dual action – immediate vasodilation plus long‑term muscle maintenance – allows brands to create products that address both acute performance and recovery, a combination rarely achieved with single‑mode ingredients.
For procurement teams, this directly translates to a lower risk of formula obsolescence. An ingredient that works through validated physiological pathways is easier to position under structure/function claims (e.g., “supports blood flow during exercise”) without overstepping regulatory boundaries. This mechanistic clarity means R&D teams can confidently shorten prototype iteration cycles, directly accelerating time‑to‑market for differentiated sports nutrition products.
Clinical Evidence & Formulation Relevance: Moving Beyond Anecdotal Claims
Scientific validation is non‑negotiable for serious supplement brands. The most cited randomised controlled trial on AAKG enrolled 35 resistance‑trained men, administering 12 g/day (4 g three times daily) for eight weeks. The AAKG group showed significant improvements in 1‑repetition‑maximum bench press and Wingate peak power compared to placebo (P < 0.05), while maintaining an excellent safety profile (doi: 10.1016/j.nut.2006.06.003). These measurable endpoints – strength and anaerobic power – are exactly what formulators need to support performance‑oriented label claims. The same trial also confirmed that AAKG does not negatively affect body composition or aerobic capacity, making it a clean addition to pre‑workout and intra‑workout products.
Key takeaways for R&D teams:
- Effective daily dose range: 10–12 g/day (split into two or three servings) replicates the positive outcomes seen in clinical studies.
- Onset of effect: Unlike acute single doses that may not produce immediate ergogenic benefits, consistent use over 4–8 weeks delivers strength and power gains.
- Safety window: The 12 g/day regimen was well tolerated; gastrointestinal side effects, often associated with free‑form arginine, were minimal due to the buffered nature of AAKG 2:1.
For brands sourcing bulk AAKG 2:1, having a COA that verifies the 2:1 molar ratio and L‑arginine content (65.5%–69.0%) is essential to match the clinical study conditions. A certified AAKG 2:1 powder eliminates the guesswork and ensures your finished product replicates the evidence‑based outcomes.
Formulation in Practice: Stability, Synergy & Delivery Solutions
AAKG 2:1 powder is a formulator‑friendly ingredient because of its near‑neutral pH (5.5–7.0) and excellent water solubility. However, professional handling requires attention to three practical aspects:
- Hygroscopicity: AAKG attracts moisture. In production, keep the relative humidity below 45% and use sealed, foil‑lined packaging. Once opened, the material should be used within 60 days to prevent clumping and maintain flowability.
- Thermal stability: The compound remains stable at standard processing temperatures (≤ 40 °C), making it suitable for dry powder blends, stick packs, and capsules. Avoid high‑shear wet granulation unless temperature is strictly controlled.
- pH compatibility: Because 2:1 AAKG is mildly acidic to neutral, it blends seamlessly with creatine monohydrate, beta‑alanine, and citrulline malate – the classic pre‑workout stack.
Synergy example: Co‑ingestion with creatine has been shown to further enhance upper‑body muscle endurance (Little et al., 2008). For a ready‑to‑mix powder, a typical combination might look like:
| Ingredient | Typical Dose per Serving | Primary Benefit |
|---|---|---|
| AAKG 2:1 | 4 g | Vasodilation, strength support |
| Creatine Monohydrate Powder | 3–5 g | Power output, muscle volumisation |
| Pure Beta-Alanine Powder | 1.6–3.2 g | Buffering, reduced fatigue |
From a strategic formulation perspective, using AAKG 2:1 in a powder format eliminates the need for complex encapsulation or coating, reducing production lead time and cost. For brands aiming to launch a “clean label” pre‑workout, this ingredient’s straightforward solubility profile means you can avoid unnecessary excipients, aligning with consumer demand for minimal ingredient decks.
Quality Benchmarks & Supply Chain Confidence
When evaluating an AAKG 2:1 supplier, professional buyers look beyond the purity number. A truly premium AAKG 2:1 powder is defined by four verifiable attributes:
- Batch‑specific COA that reports assay (≥98.0% by HPLC), L‑arginine content (65.5–69.0%), heavy metals (Pb ≤3 ppm, As ≤1 ppm, Cd ≤1 ppm, Hg ≤0.1 ppm), and microbial limits (TAMC ≤1000 CFU/g, absence of E. coli, Salmonella, S. aureus).
- Compliance with USP standards for residual solvents and identity testing, providing a globally recognised quality framework.
- Physical consistency – particle size where ≥95% passes through 80 mesh ensures uniform blending and avoids segregation in final mixes.
- Traceable sourcing from GMP‑certified facilities, with clear documentation of the synthesis route (chemical synthesis from L‑arginine and alpha‑ketoglutarate).
For procurement teams, this directly translates to lower risk of batch rejection and smoother regulatory filing. A supplier that routinely provides a complete COA and retains retention samples for at least two years offers a level of accountability that protects your brand’s supply chain. Additionally, because AAKG 2:1 is a synthetic ingredient, it avoids common allergens (soy, gluten, dairy) and delivers consistent purity regardless of seasonal crop variations – a clear advantage over some plant‑derived amino acid sources. Adopting a supplier that provides full heavy metals speciation and residual solvents data reduces the total cost of ownership by preventing costly customs holds or finished product recalls.
It is worth noting that while the FDA does not require a New Dietary Ingredient (NDI) notification for L‑arginine or its salts (due to their long history of use), responsible brands still demand full documentation. A professional AAKG 2:1 distributor will provide not only the COA but also a technical datasheet, allergen statement, and GMP declaration – all of which accelerate your compliance review for markets such as the US and EU. From a risk mitigation standpoint, verifying these documents before scale‑up eliminates the hidden cost of last‑minute supplier disqualification.
Next Steps: Validate Your Formulation with a Complimentary Sample
You now have the clinical rationale, formulation guidelines, and quality benchmarks for AAKG 2:1 powder. The next step is to experience the material’s physical behaviour and integration into your own prototype. Oclean Nutra offers a complimentary 10–20 g sample of certified AAKG 2:1 powder, accompanied by the batch‑specific COA and technical dossier. This allows your R&D team to conduct solubility tests, compatibility studies, and sensory evaluations without any upfront investment. Request your free sample and technical documentation today – responses to qualified inquiries are provided within 24 working hours.
Frequently Asked Questions
Share this product
Related Products
Ready to get started?
Contact our team for technical specifications, pricing, and customized solutions.