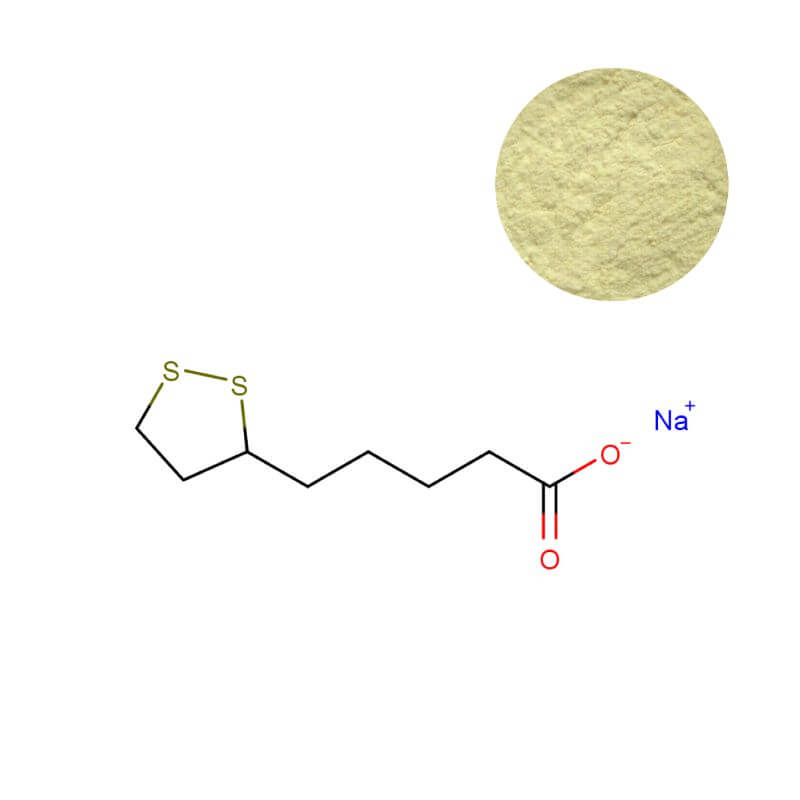
Sodium R-Lipoic Acid Powder
| Product Name | Sodium R-Lipoic Acid Powder |
| CAS Number | 176110-81-9 |
| Appearance | Pale yellowish crystalline powder |
| Purity | ≥ 99.0% (HPLC) |
| R-ALA Content | ≥ 80.0% (External Standard) |
| Packaging | 1 kg/bag, 5 kg/bag, 25 kg/drum |
| MOQ | 1 kg |
Sodium R-Lipoic Acid: Enabling Stable, High-Bioavailability Antioxidant Formulations for Sports & Healthy Aging
Formulators today face a persistent challenge: delivering potent antioxidants that survive processing, remain stable on the shelf, and actually reach target tissues. Standard R-lipoic acid, despite its well-documented benefits, suffers from poor water solubility and a tendency to polymerize, leading to variable bioavailability and short half-lives. Sodium R-Lipoic Acid addresses these limitations directly. This highly pure (
The R‑Enantiomer Advantage: Why Configuration Defines Biological Value
For a brand building a cognitive or metabolic health platform, understanding stereochemistry directly impacts product credibility. The physiological activity of lipoic acid is stereospecific—only the R‑enantiomer occurs naturally and functions as an essential cofactor in mitochondrial dehydrogenase complexes. In contrast, synthetic R,S‑lipoic acid (the racemic mixture) contains 50% of the non‑native S‑form, which may compete for transport and offer no additional benefit. Sodium R‑Lipoic Acid delivers the pure active enantiomer, ensuring that every milligram contributes to targeted pathways. Its primary mechanism involves direct radical quenching and, more importantly, activation of the Nrf2‑ARE pathway, upregulating endogenous antioxidant enzymes such as heme oxygenase‑1 and glutathione peroxidase (PMID: 18069903).
A pivotal randomized crossover trial directly compared pure R‑lipoic acid with R,S‑lipoic acid in patients with progressive multiple sclerosis. At doses yielding equivalent R‑lipoic acid plasma exposure, the R‑form produced significantly fewer gastrointestinal side effects (31 vs. 60 adverse events; P = 0.025) (doi: 10.1002/jcph.1605). This translates into a practical advantage for finished products: the ability to deliver clinically relevant doses (typically 500–600 mg/day) with better consumer compliance and fewer tolerability complaints. For brands aiming to formulate high‑potency cognitive or metabolic support supplements, choosing the R‑enantiomer is not a theoretical nuance—it directly influences end‑user acceptance and long‑term adherence. When sourcing bulk Sodium R‑Lipoic Acid, verifying enantiomeric purity through specific optical rotation data should be a standard part of supplier qualification.
Beyond Purity: The Critical Quality Markers for Sodium R‑Lipoic Acid
While a high purity percentage is expected from any reputable supplier, two additional parameters define the true quality of Sodium R‑Lipoic Acid for B2B buyers:
- Active R‑ALA content by external standard (≥80.0%) – This measurement confirms the amount of intact, biologically active R‑lipoic acid after reconstitution. It is a more meaningful indicator of functional potency than simple area‑normalization purity, which may include degradation products or the S‑enantiomer. From a formulation standpoint, this figure directly supports the label claims your brand will make.
- Specific optical rotation ([α]²⁵ᴰ +70.0° to +90.0°) – A direct measure of enantiomeric integrity, this test verifies that the material consists almost entirely of the desired R‑form. Values outside this range signal racemization or contamination with the inactive S‑isomer.
From a compliance standpoint, these parameters are routinely documented in batch‑specific Certificates of Analysis. A premium Sodium R‑Lipoic Acid supplier will provide these data for every lot, enabling brands to include them in their own quality filings and ensuring consistency across production runs. Additionally, microbiological limits (total plate count ≤1,000 CFU/g, absence of pathogens) and heavy metals (Pb ≤1.0 mg/kg, inorganic As ≤1.0 mg/kg) are verified according to USP methods, safeguarding final product safety without requiring costly re‑testing by the finished‑goods manufacturer. For procurement teams, a complete COA package reduces redundant qualification work and accelerates time to market.
Formulation in Practice: Leveraging Water Solubility for New Delivery Systems
The conversion of R‑lipoic acid to its sodium salt fundamentally changes its physicochemical profile. Sodium R‑Lipoic Acid is freely water‑soluble, unlike the parent compound, and exhibits markedly improved stability against heat‑induced polymerization. For a brand's R&D team, this opens formulation possibilities that were previously impractical:
- Liquid and semi‑solid formats – Ready‑to‑drink shots, stick packs, and liquid softgels can now incorporate R‑lipoic acid without precipitation or rapid degradation. The water‑soluble nature allows for clear, homogeneous solutions—a critical aesthetic for premium beverage products.
- Synergistic combinations – It pairs particularly well with other mitochondrial nutrients. A common strategy is to combine it with Alpha Lipoic Acid Powder in a dual‑delivery system: the sodium salt provides rapid aqueous‑phase activity, while the free acid suits oil‑based or controlled‑release blends. For comprehensive mitochondrial support, many formulators also layer in Acetyl L-Carnitine or CoQ10 Ubiquinone.
| Delivery Format | Typical Inclusion Level (per serving) | Key Formulation Consideration |
|---|---|---|
| Powdered beverages / stick packs | 100–200 mg | Use direct blending; avoid excessive moisture during mixing (control relative humidity below 45%). |
| Hard capsules / tablets | 200–300 mg | Blend with flow aids; sodium salt is less prone to sticking than free acid. |
| Liquid shots / softgels | 50–150 mg | Dissolve in aqueous phase; protect from light with opaque packaging. |
In practice, the enhanced stability of the sodium salt reduces the need for complex encapsulation technologies, lowering both development costs and time to market. For brands targeting athletes or active lifestyle consumers, this ingredient allows the creation of clean‑label products with minimal excipients while maintaining potency through the stated shelf life (typically 24 months when stored cool and dry). The formulation flexibility it offers can be the difference between a generic product and an innovative delivery format that captures consumer attention.
Risk Mitigation: A Sourcing Checklist for Sodium R‑Lipoic Acid
Selecting a supplier for this specialized ingredient requires scrutiny beyond standard vendor qualification. Because optical purity is not visible in routine HPLC area‑percent reports, buyers must explicitly verify that the material meets both the total purity and the active R‑ALA content specifications. A disciplined procurement process—one that looks beyond price per kilogram—can prevent downstream failures. The following checklist outlines objective criteria for evaluating potential partners:
- Batch‑specific COA including: total purity (HPLC area %), active R‑ALA content (external standard), specific optical rotation, residual solvents (USP <467>), and full heavy metals panel.
- Third‑party testing confirmation for at least one lot annually, verifying enantiomeric purity by chiral HPLC.
- GMP compliance documentation for the manufacturing site (even if the trading partner does not manufacture, they should source from GMP‑certified facilities).
- Traceability – the ability to provide a complete chain of custody from raw material to finished product.
- Supply chain transparency – details on sourcing of the synthetic precursor and any steps taken to avoid cross‑contamination.
From a total‑cost‑of‑ownership perspective, paying a modest premium for a fully documented, consistently pure Sodium R‑Lipoic Acid eliminates the risk of batch failures, regulatory delays, or consumer complaints. It also streamlines the NDI notification or novel food dossier preparation, as the technical data package is already aligned with international standards. The few minutes spent verifying these points before placing an order can save months of troubleshooting later.
Next Steps for Your Product Development
Integrating Sodium R‑Lipoic Acid into your next formulation offers a clear path to differentiate products in the crowded antioxidant and sports nutrition categories. Its combination of verified R‑enantiomer activity, water solubility, and thermal stability addresses the most common technical hurdles associated with lipoic acid. To move from concept to commercial reality, the first step is to evaluate the material firsthand and access the supporting documentation that will underpin your regulatory filings.
Request your complimentary sample and complete technical dossier – including a batch‑specific COA, stability summary, and formulation guide – and accelerate your path to a market‑ready product backed by uncompromising quality.
Frequently Asked Questions
Share this product
Related Products
Ready to get started?
Contact our team for technical specifications, pricing, and customized solutions.