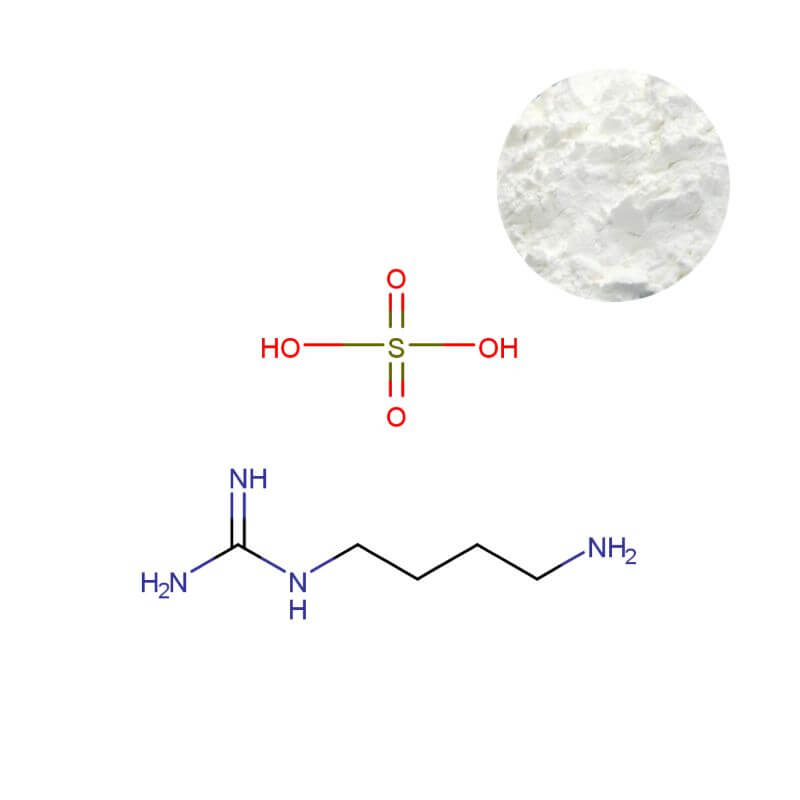
Agmatine Sulfate Powder
| Product Name | Agmatine Sulfate Powder |
| CAS Number | 2482-00-0 |
| Appearance | White or off-white crystalline powder |
| Purity | ≥ 99.0% (by HPLC) |
| Packaging | 1 kg/bag, 5 kg/bag, 25 kg/cardboard drum |
| MOQ | 1 kg |
Agmatine Sulfate: A Precision Ingredient for Neurohealth & Sports Nutrition Formulations
For brands developing next-generation cognitive health, mood support, or pre-workout products, the challenge often lies in identifying ingredients that offer both clinical substantiation and formulation practicality. Agmatine Sulfate, a metabolite of L-arginine, has emerged as a distinct solution supported by recent human clinical data and a well-defined safety profile. A premium-grade ingredient standardized to ≥99.0% purity and verified by HPLC provides the batch-to-batch consistency essential for regulatory filing and reliable product performance. This article outlines the science, quality benchmarks, and practical formulation strategies that define a sophisticated approach to incorporating this unique ingredient into your product portfolio.
Mechanism of Action: The Dual-Pathway Approach to Neuromodulation & Vascular Support
For formulators, the value of Agmatine Sulfate lies in its dual mechanism, which operates through two distinct physiological pathways. Unlike single-target ingredients, agmatine functions as both an NMDA receptor antagonist and a modulator of nitric oxide synthase (NOS). This unique profile enables it to support a broad spectrum of benefits, from neuroprotection to enhanced vascular function. A 2020 open-label clinical study provided compelling human data, demonstrating that oral agmatine sulfate (at 2.67g/day over two months) significantly reduced pain intensity by 46.4% (p < 0.00001) in patients with small fiber neuropathy (doi: 10.3390/nu12020576).
For the product developer, this translates into a scientifically grounded foundation for formulations targeting:
- Cognitive & Mood Health: NMDA receptor modulation supports neural homeostasis.
- Post-Exercise Recovery: Its role in the NO pathway may help manage exercise-induced discomfort.
- General Neuroprotection: Helps maintain neuronal resilience against excitotoxicity.
Practically speaking, this dual-pathway nature allows brands to position products not just for a single benefit, but for a synergistic effect on both mental and physical well-being—a compelling value proposition in today’s integrated health market. From a commercial perspective, this versatility enables a single ingredient to serve multiple product lines, from pre-workout to mood support, streamlining inventory management and reducing supply chain complexity.
Quality Benchmarks: What to Look for in a Premium Agmatine Sulfate Ingredient
For sourcing teams evaluating bulk Agmatine Sulfate, relying on a simple “≥99%” purity claim is insufficient. A truly premium ingredient is defined by a comprehensive specification that ensures both safety and functional performance. Key sourcing considerations should include a detailed Certificate of Analysis (COA) that confirms critical parameters beyond the primary assay—especially important for a synthetic ingredient where process control dictates final quality.
A robust quality profile, such as that expected from established suppliers, typically features:
- Assay (HPLC): ≥99.0% – ensuring active ingredient potency.
- Particle Size: ≥95% through 80 mesh – crucial for flowability in capsule filling and dissolution in drink mixes.
- Residue on Ignition: ≤0.1% – a critical measure of inorganic impurities, reflecting synthetic purity.
- Sulfate Content (Titration): 42.5% – 44.5% – verifying the correct salt form (monosulfate) and preventing contamination from other salts.
- Regulatory Identifiers: CAS #2482-00-0 and FDA UNII #RU0176QL8I — ensuring global traceability and seamless regulatory filing.
A lesser-known but critical quality metric is the verification of sulfate content. Professional R&D teams use this to confirm the ingredient is the stable, single-salt form of agmatine, rather than a mixture of free base and other salts. This level of detail, provided in a fully traceable COA, directly reduces formulation risk and supports a brand’s claims for product purity and consistency. For brands targeting premium market segments, these granular specifications serve as tangible differentiators that can be highlighted in consumer-facing marketing materials.
Beyond basic assay purity, strategic procurement should evaluate the specific synthesis route. While multiple methods achieve ≥99.0%, advanced chemical synthesis or enzymatic pathways often yield a superior residual solvent profile. For brands targeting the “Clean Label” segment or third-party certifications like NSF, this technical nuance is a critical differentiator that ensures regulatory compliance and consumer trust.
Formulation in Practice: Stability, Synergy, and Delivery Strategies
From a product development risk management standpoint, understanding how an ingredient behaves during manufacturing is as critical as its clinical profile. Successfully integrating Agmatine Sulfate into finished products requires addressing its physical properties and optimizing its compatibility with other ingredients. One of the primary considerations is its hygroscopic nature. In a production environment, maintaining relative humidity below 60% during blending and encapsulation is a practical step to prevent clumping and ensure uniform dosing—a detail that directly impacts manufacturing yield and final product consistency. For encapsulation, HPMC (cellulose) shells are often preferred over traditional gelatin to mitigate potential shell brittleness caused by moisture migration over the product’s 24-month shelf life.
| Formulation Consideration | Strategic Approach | Commercial Implication |
|---|---|---|
| Hygroscopicity & Stability | Store in original sealed packaging; use desiccants in final packaging. Formulate in low-humidity environments. | Prevents caking and degradation, ensuring product shelf-life and consumer experience. For brands, this translates to lower return rates and stronger brand trust. |
| Synergistic Combinations | Pair with L-Citrulline for enhanced NO pathway support in pre-workouts. Combine with Alpha Lipoic Acid for neuropathic support formulas. | Creates differentiated, multi-modal products that address complex consumer needs, enabling premium pricing strategies. |
| Dosage & Delivery | Typical effective daily dose ranges from 500mg to 2,670mg, based on clinical research. Capsules, tablets, and drink powders are all viable delivery formats. | Flexibility in dosage and format allows for targeting specific price points and consumer segments, from value-oriented to premium functional beverages. |
An effective formulation strategy also leverages known synergies. For instance, combining Agmatine Sulfate with ingredients that target the same pathways—such as L-Citrulline for NO support—can create a more potent, multi-faceted effect. This approach allows brands to build complex, scientifically grounded stacks that are difficult for competitors to replicate, directly contributing to product lifecycle longevity.
Mitigating Risk: A Framework for Sourcing a Certified Agmatine Sulfate Supplier
Selecting a supply partner for a critical ingredient like Agmatine Sulfate requires a risk-mitigation mindset. The goal is not just to procure a raw material, but to secure a consistent, compliant, and traceable supply chain. A robust safety profile is foundational; recent 2024 studies confirm that the ingredient is non-mutagenic and non-genotoxic under the tested conditions, providing crucial data for regulatory submissions (doi: 10.1016/j.toxrep.2024.101720).
For a brand’s procurement and regulatory teams, the true value of a supplier is demonstrated through their ability to provide:
- Comprehensive Documentation: A batch-specific COA that includes not just the assay, but also heavy metals (Pb, As, Cd, Hg), residual solvents, and microbiological tests.
- Supply Chain Transparency: Clear information on the origin of materials and manufacturing processes, ensuring traceability from synthesis to finished product.
- Technical Support: A partner that can provide formulation advice and stability data, reducing development time and costs.
- Regulatory Readiness: A clear understanding of the ingredient’s status (e.g., FDA UNII #RU0176QL8I) and the ability to assist with technical documentation for global market access.
This approach to supplier selection transforms a simple transaction into a strategic partnership, effectively insulating a brand from supply disruptions, quality failures, and compliance issues. From a total cost of ownership (TCO) perspective, investing in a fully documented supply chain often yields lower long-term costs by minimizing batch rejections and regulatory delays.
Next Steps for Your Product Development Program
Agmatine Sulfate represents a unique opportunity to develop differentiated products in the expanding neurohealth, mood, and sports nutrition categories. By focusing on high-quality specifications (≥99.0% purity, verified via HPLC and comprehensive testing) and partnering with a supplier that provides full technical transparency, you can confidently bring a scientifically supported ingredient to market. The most direct path from concept to compliant product is to evaluate the ingredient firsthand under your own quality protocols. Request Your Complimentary Sample & Technical Dossier to begin your risk-free evaluation and accelerate your development timeline.
Frequently Asked Questions
Share this product
Related Products
Ready to get started?
Contact our team for technical specifications, pricing, and customized solutions.