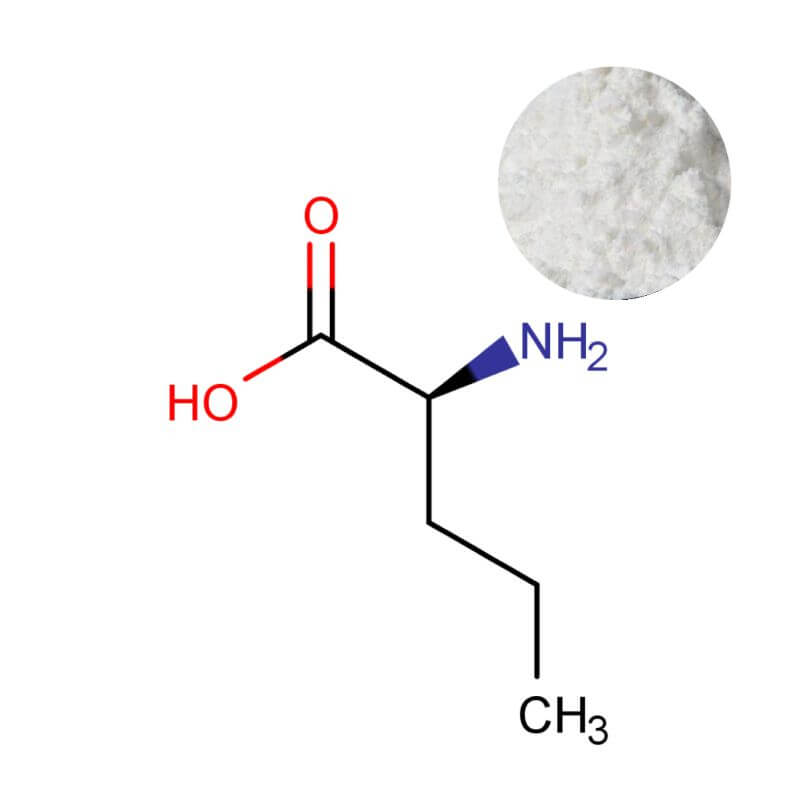
L-Norvaline Powder
| Product Name | L-Norvaline Powder |
| CAS Number | 6600-40-4 |
| Appearance | White crystalline powder |
| Purity | 99% min. (by HPLC) |
| Packaging | 1 kg/foil bag, 25 kg/paper drum |
| MOQ | 1 kg |
L-Norvaline Powder: A Clinically-Supported Ingredient for Sports & Cognitive Health Formulations
For brands aiming to differentiate their next-generation formulations, relying on generic vasodilators or basic nootropic stacks no longer guarantees market impact. The modern consumer—whether an athlete seeking enhanced endurance or an aging adult prioritizing mental sharpness—demands ingredients with clear, scientifically-validated mechanisms. L-Norvaline, a non-protein amino acid and potent arginase inhibitor, addresses this need directly. Standardized to a minimum of 99% purity by HPLC, this ingredient offers formulators a reliable tool for modulating nitric oxide (NO) bioavailability. Beyond the specification sheet, the value of L-Norvaline lies in its demonstrated potential in both sports and cognitive applications. Understanding its quality benchmarks and formulation nuances allows brands to confidently position products in premium segments where efficacy claims must be substantiated. The following sections outline the science behind this ingredient, the quality benchmarks that ensure its efficacy, practical formulation strategies, and the critical supply-chain considerations that mitigate risk for your product development projects.
Targeting Nitric Oxide Bioavailability: The Arginase Inhibition Mechanism
The primary physiological role of L-Norvaline is its action as a competitive, non-selective inhibitor of the arginase enzyme. Arginase competes with nitric oxide synthase (NOS) for their common substrate, L-arginine. By inhibiting arginase, L-Norvaline effectively shifts the balance toward the NOS pathway, increasing the production of nitric oxide. This biochemical shift has two major downstream effects relevant to supplement formulators:
- Vascular support: Enhanced NO promotes vasodilation, improving blood flow, nutrient delivery, and oxygen transport—a key mechanism for endurance and muscle pump formulations (doi: 10.1007/s13311-018-0668-6).
- Neuronal protection and plasticity: Beyond the vascular role, preclinical work has revealed that L-Norvaline can reduce neuroinflammation and beta-amyloid burden, while increasing markers of synaptic health such as PSD95 (doi: 10.4103/1673-5374.255980).
What does this mean for your product portfolio? It positions L-Norvaline as a dual-action ingredient: in sports nutrition, it supports the physiological endpoints athletes care about (endurance, recovery); in cognitive health, it targets mechanisms beyond simple blood flow—addressing inflammation and synaptic integrity. Practically speaking, for brand managers, this dual mechanism translates directly into the ability to build two distinct product lines from a single, well-documented ingredient, streamlining raw material qualification and inventory management, thereby reducing complexity and improving supply chain efficiency.
Defining Quality: Beyond 99% Purity to Optical Rotation and Batch Consistency
When sourcing bulk L-Norvaline, a certificate of analysis (COA) stating “≥99% purity” is expected, but it is not sufficient. Two additional parameters separate premium-grade material from commodity-grade alternatives:
- Specific optical rotation [α]²⁰/ᴰ: Because L-Norvaline exists as a chiral molecule, verifying the correct L-isomer is essential. The accepted range of +23.0° to +26.0° (c=10, 6N HCl) confirms that the product is the biologically active form. An off-spec rotation could indicate the presence of the D-isomer or other chiral impurities, directly impacting efficacy.
- Heavy metal limits: Reputable suppliers test for elemental impurities well below pharmacopeial limits (e.g., Pb ≤1.0 mg/kg, As ≤1.0 mg/kg, Cd ≤0.5 mg/kg, Hg ≤0.1 mg/kg). This not only satisfies global regulatory requirements but also protects your brand from the long-term reputational risk of contamination.
From a risk-mitigation standpoint, the presence of these data points on a batch-specific COA is non-negotiable. They ensure that every kilogram of L-Norvaline you receive will perform identically in your finished product, avoiding costly formulation reworks or stability surprises. For procurement teams, a supplier that routinely provides full optical rotation data and heavy metals analysis demonstrates a commitment to transparency that lowers the total cost of ownership—by eliminating the hidden costs of failed quality checks and delayed market entries. The table below summarizes the critical quality attributes and their direct commercial implications:
| Quality Parameter | Specification (Typical) | Commercial Implication |
|---|---|---|
| Assay (HPLC) | ≥99.0% | Ensures potency and justifies label claims. |
| Specific Rotation | +23.0° to +26.0° | Confirms correct isomer for targeted biological activity. |
| Heavy Metals (Pb, As, Cd, Hg) | ≤1.0 / 1.0 / 0.5 / 0.1 mg/kg | Reduces regulatory rejection risk; supports global market access. |
| Particle Size | ≥95% through 80 mesh | Facilitates uniform blending in powders and capsules. |
Formulation Strategies: Synergistic Combinations and Delivery Considerations
An effective L-Norvaline formulation leverages its mechanism while addressing practical production realities. The most researched synergy is with L-Arginine HCl itself: L-Norvaline preserves arginine from being wasted by arginase, while exogenous L-arginine provides abundant substrate for NO synthesis. In preclinical models, this combination improved glycemic control and lipid profiles more robustly than either alone (doi: 10.1016/j.yexmp.2022.104763). For brands targeting cognitive applications, pairing L-Norvaline with Phosphatidylserine (PS) or Citicoline (CDP Choline) may amplify synaptic benefits, creating a differentiated product that appeals to the growing “healthy aging” demographic.
From a processing standpoint, L-Norvaline powder exhibits good flowability and stability under standard manufacturing conditions. However, two practical tips can safeguard finished product quality:
- Moisture control: While L-Norvaline is not highly hygroscopic, maintaining blending environment humidity below 45% RH prevents caking and ensures uniform content uniformity—especially important in high-concentration pre-workout powders.
- pH compatibility: In ready-to-drink (RTD) applications, avoid highly acidic formulas (pH <3) for extended periods, as extreme acidity may gradually degrade amino acids. Buffer systems or encapsulation can mitigate this risk.
The typical inclusion rate in supplements ranges from 100 mg to 300 mg per serving, based on extrapolation from animal studies and prevailing market products. It is advisable to start with the lower end for cognitive formulas and titrate upward for sports products, always verifying stability with your contract manufacturer. For brands planning to launch multiple SKUs (e.g., a pre-workout powder and a nootropic capsule), establishing a single L-Norvaline concentration across lines simplifies procurement and quality control while still allowing for formula differentiation through companion ingredients.
Securing Your Supply Chain: Transparency, COA, and Technical Support
The true cost of an ingredient is not its per-kilo price, but the total cost of ownership—including the risks of delayed launches, failed inspections, and consumer complaints. When evaluating a L-Norvaline supplier, a robust quality system is as critical as the specification sheet. Premium suppliers differentiate themselves through:
- Batch-specific documentation: Every shipment should be accompanied by a full COA covering identity, purity, optical rotation, heavy metals, and microbial counts. This supports your own incoming quality checks and regulatory filings.
- Traceability and audits: Facilities operating under GMP guidelines (e.g., FSSC 22000 or equivalent) allow for supply-chain transparency. Knowing the synthesis route (chemical or enzymatic) and the origin of starting materials can be vital for allergen and contaminant risk assessments.
- R&D support: Access to formulation expertise and accelerated stability data can cut months off your development timeline. Reliable partners typically provide technical dossiers and respond to inquiries within 24 working hours.
In practice, this means that when you request a sample of L-Norvaline, you should receive not only the powder but also a preliminary COA and a clear path to scale-up. This upfront transparency directly reduces the risk of supply interruptions and ensures that the ingredient you qualify today will match the bulk material delivered tomorrow. Experienced procurement officers view such documentation as a sign of a supplier’s operational maturity, enabling them to confidently commit to long-term sourcing agreements without fearing last-minute specification changes.
Next Steps for Your Product Development
Integrating L-Norvaline into your brand’s portfolio offers a science-backed route to differentiation in both sports nutrition and cognitive health categories. By focusing on verified quality—≥99% purity, correct optical rotation, and rigorous contaminant testing—you can build formulations that deliver consistent, marketable benefits while minimizing regulatory and manufacturing risks. The next move is straightforward: evaluate the ingredient firsthand and access the technical documentation that supports your development work.
Request your complimentary sample and comprehensive technical dossier to confirm the powder’s characteristics in your own lab and accelerate your path to a finished product that meets the highest standards of efficacy and safety. This step allows your R&D team to verify batch consistency and explore synergies with your existing formulations—turning scientific potential into tangible consumer benefits.
Frequently Asked Questions
Share this product
Related Products
Ready to get started?
Contact our team for technical specifications, pricing, and customized solutions.