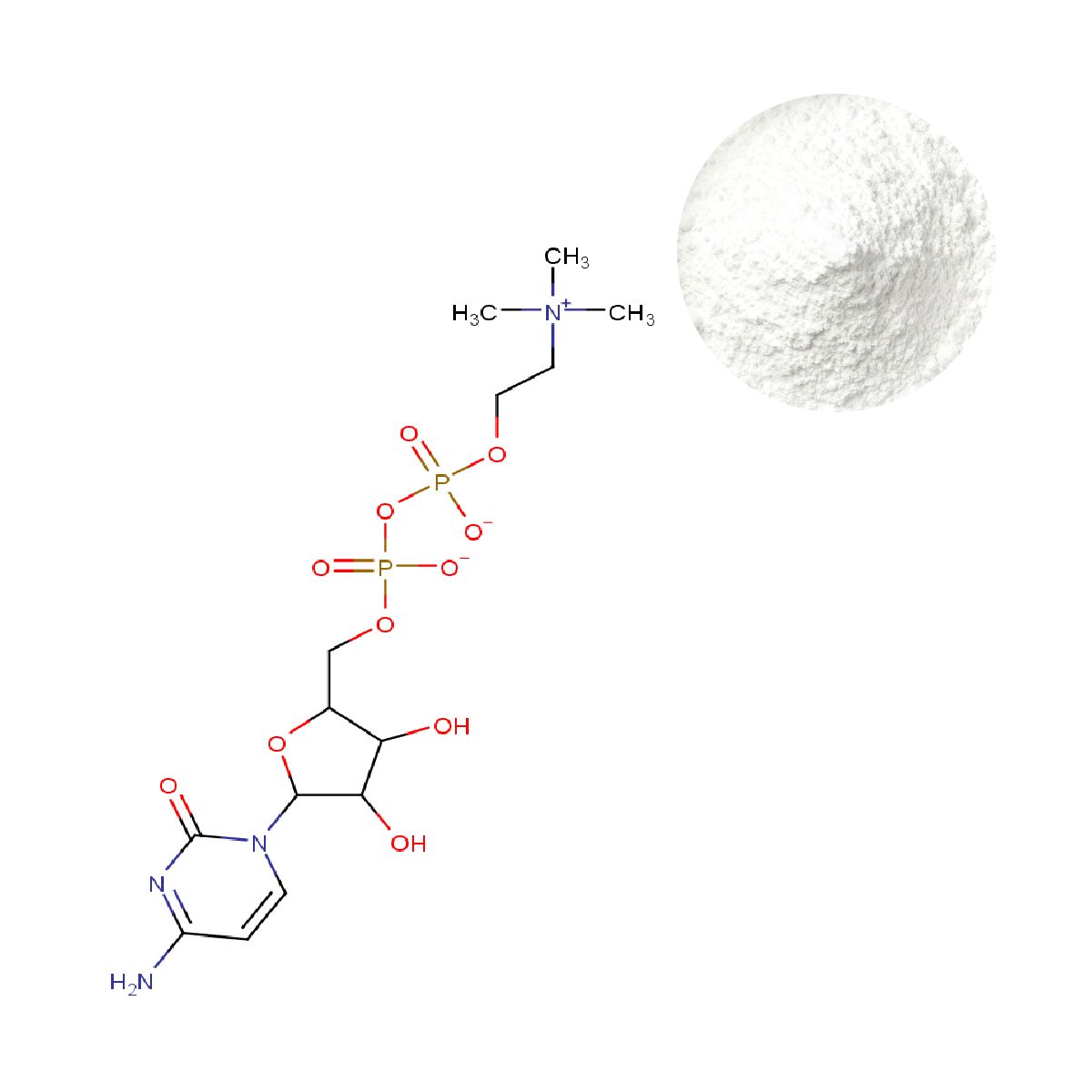
Citicoline CDP Choline Powder
| Product Name | Citicoline CDP Choline Powder |
| CAS Number | 987-78-0 |
| Appearance | White to off-white fine powder |
| Purity | ≥99% (HPLC) |
| Packaging | 1 kg/bag, 5 kg/bag, 25 kg/drum |
| MOQ | 1 kg |
Citicoline CDP Choline Powder: A Clinically‑Validated Foundation for Cognitive Health Formulations
For brands developing next‑generation nootropic and healthy aging products, differentiating through credible science is no longer optional—it is the entry ticket. While the cognitive category is crowded with stimulants and vague “brain boosters”, formulators are increasingly seeking ingredients that are endogenously relevant and backed by human data. Citicoline (CDP‑choline)—an intermediate in the Kennedy pathway, the sole route for neuronal membrane phospholipid synthesis—meets this demand. Verified by HPLC at ≥99% purity and characterized by a tight particle size distribution (≥95% through 80 mesh), a premium citicoline powder delivers the batch‑to‑batch consistency essential for both clinical reproducibility and manufacturing efficiency. For procurement and R&D leaders, this translates directly into predictable finished‑product performance and a defensible position in an increasingly evidence‑driven market. This article translates recent clinical evidence, regulatory boundaries, and formulation realities into a practical roadmap for successfully bringing a citicoline‑based product from concept to market—without overclaim or compliance surprises.
Endogenous Intelligence: Why Citicoline’s Mechanism Matters Beyond “Choline Donation”
Citicoline is not merely a choline source; it is the only molecule that simultaneously supplies choline and cytidine in the precise ratio required for neuronal membrane biogenesis. Once absorbed, it hydrolyses to choline and cytidine. Choline supports acetylcholine synthesis and serves as a precursor for phosphatidylcholine, while cytidine is converted to uridine, a rate‑limiting substrate for synaptic membrane formation (doi: 10.14336/AD.2022.0913). This dual pathway explains why clinical effects—such as improved episodic memory in older adults—are observed at daily intakes of 500–1000 mg, rather than with choline salts alone.
What does this mean for a finished product? Unlike acetyl‑L‑carnitine or alpha‑GPC—which primarily donate acetyl or choline groups—citicoline addresses both the structural (membrane repair) and neurotransmitter (acetylcholine) components of cognitive vitality. Practically speaking, this positions it as a foundational ingredient for chronic neuroprotection and healthy aging formulas, not an acute stimulant. The 99% purity grade—consistently achievable through controlled fermentation—ensures that every batch delivers the full spectrum of endogenous metabolites without the variability inherent in botanical extracts. For brands, this predictability translates directly into more reproducible clinical outcomes, stronger intellectual property protection for proprietary blends, and a cleaner label story that resonates with science‑literate consumers.
Quality Benchmarks: Translating Analytical Chemistry into Commercial Certainty
In bulk citicoline procurement, the difference between a reliable ingredient and a formulation failure often lies in three frequently overlooked parameters: pH, 5′‑CMP content, and particle morphology. A premium citicoline CDP choline powder typically exhibits a pH of 2.5–3.5 (1% aqueous solution) due to its inner salt form; this acidity, while compatible with most capsules and tablets, requires careful evaluation when co‑formulated with alkaline minerals or enteric coatings. The related substance 5′‑cytidine monophosphate (5′‑CMP) is the primary impurity and is specified at ≤1.0%—a critical control point because elevated 5′‑CMP indicates hydrolysis during processing and correlates with reduced stability. Particle size (≥95% through 80 mesh) is not merely a flowability metric: uniform fine particles minimise segregation in high‑speed direct compression and ensure rapid dissolution in stick packs. Experienced procurement teams therefore treat these three metrics as non‑negotiable gatekeepers when qualifying new suppliers.
From a compliance standpoint, citicoline’s regulatory status varies significantly by region. In the United States, the ingredient is self‑affirmed GRAS for dietary supplements; no NDI notifications have been filed, and its long history of safe use is well documented. The European Union authorises citicoline as a Novel Food for specified non‑beverage products, yet a 2024 EFSA opinion concluded that a cause‑and‑effect relationship for “memory function” claims has not been established (doi: 10.2903/j.efsa.2024.8861). The strategic implication is clear: while structure‑function claims on “neuronal membrane integrity” or “acetylcholine synthesis” remain permissible globally, explicit “memory improvement” messaging is currently off‑limits in the EU. A GMP‑certified supplier who provides full disclosure of residual solvents, heavy metals (Pb ≤0.5, As ≤0.5, Cd ≤0.3, Hg ≤0.1 mg/kg), and microbiological purity (absent pathogens) is not a commodity vendor—it is a partner that de‑risks your regulatory pathway. For brands targeting EU entry, verifying that your supplier can transfer the complete Novel Food dossier is as critical as the ingredient price itself.
Formulation in Practice: Stability, Compatibility, and Synergy
For formulation scientists racing to commercialize citicoline‑based products, the intersection of raw‑material properties and process controls is where development timelines are won or lost. Citicoline’s water solubility (freely soluble) simplifies incorporation into powder blends, but its hygroscopic nature demands manufacturing discipline. An effective formulation protocol requires maintaining blending‑room humidity below 40% RH; under such conditions, the powder remains free‑flowing and does not cake. This practical insight often proves more decisive than packaging selection—although aluminum foil bags with double PE liners are mandatory for long‑term storage. Accelerated stability studies (40 °C/75 % RH for six months) demonstrate that properly packaged citicoline retains >98% potency, affording a 24‑month shelf life at ambient conditions.
For tablet and two‑piece hard capsule applications, direct compression is feasible when the blend contains adequate lubricants (e.g., magnesium stearate) and disintegrants. Because citicoline is acidic, it should not be granulated with carbonate‑based excipients. Pre‑gelatinised starch or microcrystalline cellulose are preferred fillers. A particularly effective synergy exists with phosphatidylserine (PS): citicoline supplies the choline and cytidine for new membrane synthesis, while PS facilitates membrane fluidity and neuron signalling. From a portfolio strategy perspective, this combination is increasingly adopted in premium cognitive‑aging products because it addresses both membrane architecture and neurotransmitter kinetics—a distinction that resonates with informed consumers and justifies a higher price point.
It is worth noting that while systematic reviews confirm positive effects on cognition in mild cognitive impairment and dementia, the overall quality of older trials has been criticised (doi: 10.3390/nu15020386). For the modern formulator, this underscores the importance of using only high‑purity, batch‑standardised citicoline (≥99% HPLC purity) when seeking to replicate outcomes of well‑controlled recent studies (e.g., Nakazaki 2021). Consistency, not merely purity, is the true driver of clinical reproducibility—and therefore the true driver of brand trust.
Mitigating Risk: The Checklist for Sourcing Premium Citicoline
Selecting a citicoline supplier involves far more than price per kilogram. Based on audits of leading global supply chains, the following five criteria are predictive of a low‑risk, high‑value partnership:
| Due Diligence Item | Why It Matters |
|---|---|
| Full‑panel Certificate of Analysis (CoA) | Beyond assay and heavy metals, CoA must include 5′‑CMP, pH, particle size, and microbial limits. A supplier who cannot provide these per batch is not equipped for regulated markets. |
| GMP compliance with audit trail | Verifiable GMP certification (e.g., FSSC 22000, ISO 22000) and acceptance of third‑party facility audits ensure that process controls—not just final testing—guarantee quality. |
| Regulatory dossier availability | For EU entry, Novel Food authorisation documentation must be transferable; for the US, a GRAS determination file should be available for review by your legal team. |
| Stability‑backed shelf life | 24‑month shelf life claims should be supported by real‑time or accelerated studies conducted on the specific purity grade you purchase—not generic literature data. |
| Supply chain transparency | Knowledge of the fermentation site, purification steps, and solvent residues (compliant with USP <467>) is essential for both allergen declarations and sustainability reporting. |
Applying this checklist transforms procurement from a transactional activity into a strategic risk‑mitigation exercise. The total cost of ownership for a certified, pure, and fully documented citicoline is often lower than that of an apparently cheaper alternative, when factoring in rejected batches, reformulation delays, and lost market opportunities. A distributor who curates such material—and provides formulation support—effectively acts as an extension of your R&D team. This is why sophisticated sourcing organisations increasingly prioritise technical competence over spot‑price advantages.
Next Steps for Your Product Development
Citicoline CDP choline powder, when specified at ≥99% purity and backed by rigorous analytical and regulatory documentation, offers a scientifically grounded route to differentiate cognitive health products. The ingredient’s dual mechanism, compatibility with modern manufacturing, and mature supply base make it suitable for both rapid commercialisation and long‑term brand building. To translate this scientific foundation into a commercial advantage with minimal regulatory friction, a hands‑on evaluation is the most efficient first step. A representative sample and complete technical dossier—including batch‑specific CoA, stability summary, and regional compliance statements—enable your internal teams to verify quality, test formulation fit, and accelerate your timeline with full confidence.
Frequently Asked Questions
Share this product
Related Products
Ready to get started?
Contact our team for technical specifications, pricing, and customized solutions.