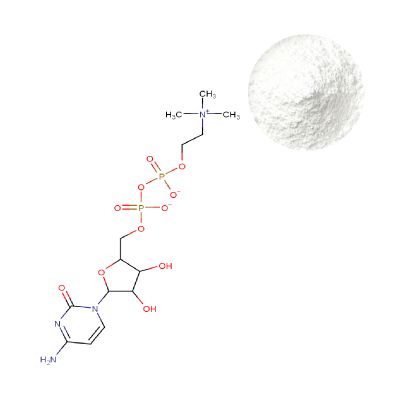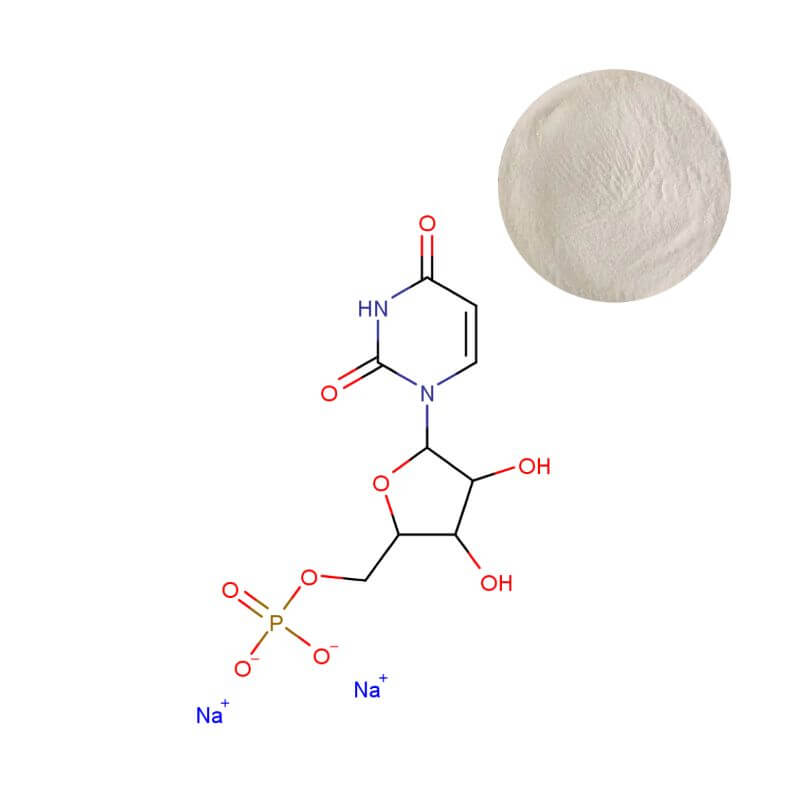
Uridine Monophosphate Disodium Powder
| Product Name | Uridine 5'-Monophosphate Disodium Powder |
| CAS Number | 3387-36-8 |
| Appearance | White to off-white crystalline powder |
| Purity | ≥98% or ≥99% (by HPLC) |
| Packaging | 1 kg/bag, 5 kg/bag, 25 kg/drum |
| MOQ | 1 kg |
Uridine 5'-Monophosphate Disodium: A Strategic Ingredient for Cognitive Vitality and Cardiometabolic Resilience
In today's competitive landscape of cognitive health products, brands seeking to differentiate must look beyond generic blends and invest in ingredients with robust mechanistic evidence and formulation versatility. Uridine 5'-Monophosphate Disodium (UMP), a key nucleotide involved in membrane phospholipid synthesis and cellular energy metabolism, offers precisely that—a science‑backed foundation for products targeting memory, neuroprotection, and even cardiac function. Premium Uridine 5'-Monophosphate Disodium powder, typically supplied at ≥98% or ≥99% purity (HPLC-verified), is not just a raw material; it represents a strategic component that can elevate a product line's credibility and efficacy. This article provides an in‑depth analysis of the critical quality benchmarks, formulation considerations, and supply chain factors that empower R&D and procurement teams to confidently integrate UMP into innovative finished products, while also illuminating the business implications behind each technical detail.
The Dual Mechanism of UMP: From Synaptic Integrity to Mitochondrial Protection
For product developers, understanding UMP's dual mechanism is key to crafting differentiated health claims. Uridine 5'-Monophosphate Disodium exerts its physiological effects through two well‑characterised pathways. First, as a precursor to UTP and CTP, it fuels the Kennedy pathway for phosphatidylcholine synthesis—the primary phospholipid of neuronal membranes. This process directly supports synaptic plasticity and neurotransmitter release. A pivotal randomised, double‑blind, placebo‑controlled trial in 99 healthy middle‑aged adults demonstrated that daily supplementation with 600 mg of 5'‑UMP for 12 weeks significantly improved composite memory scores (Cognitrax assessment), with particular benefit in subjects reporting subjective memory decline (UMIN‑CTR ID: UMIN000045507). This human evidence substantiates UMP's role in formulations targeting age‑related cognitive maintenance, and importantly, it provides a quantifiable endpoint for marketing claims.
Second, UMP contributes to mitochondrial ATP‑sensitive potassium channel (mitoKATP) activation via its metabolite UDP. Preclinical research using a rat coronary occlusion model revealed that intravenous UMP (30 mg/kg) reduced the ischemic alteration index by 3.5‑fold, decreased ventricular premature beats by 5.6‑fold, and shortened ventricular tachycardia duration by 9.4‑fold—effects that were abolished by a selective mitoKATP blocker (doi: 10.1016/j.exger.2006.03.005). It is critical to note that these findings, while mechanistically informative, are derived from animal models and should be referenced strictly as mechanistic background; they cannot be extrapolated to human disease claims. For brands, this distinction means the cardioprotective narrative must be framed around foundational cellular energy support—such as "helps maintain healthy energy metabolism during physical activity"—rather than any suggestion of cardiac treatment or disease mitigation. From a product development standpoint, this dual mechanism enables brands to position UMP‑based products not merely as cognitive enhancers, but as holistic vitality ingredients that support both mind and cardiovascular resilience—a compelling narrative for premium positioning.
Quality Benchmarks: Beyond Purity to Structural Integrity and Consistency
When sourcing bulk Uridine 5'-Monophosphate Disodium, the industry standard of ≥98% or ≥99% purity (by HPLC) is only the starting point. True quality differentiation—and the assurance it provides to brand owners—lies in how that purity is maintained across batches and how the molecule's physical behaviour aligns with formulation needs. Loss on drying (≤26.0%) and specific rotation (−11.0° to −17.0°) are critical parameters that reflect the crystalline form and absence of residual solvents or moisture, directly influencing flowability and long‑term stability in finished products. Moreover, the interaction of UMP with lipid membranes—studied at a molecular level—shows that the disodium salt form (UMPDSS) can adsorb to phospholipid bilayers without disrupting their structure, while altering membrane thickness and hydration (doi: 10.1039/c9sm01257e). Practically speaking, this means that a high‑purity, structurally consistent UMP powder integrates more predictably into lipophilic matrices, reducing the risk of phase separation or altered dissolution profiles in capsule or tablet formulations—factors that directly impact consumer perception and product performance.
Key sourcing considerations for procurement and R&D teams include:
- Comprehensive COAs: Every batch should be accompanied by a Certificate of Analysis documenting not only assay by HPLC, but also residual solvents (USP <467>), heavy metals (ICP‑MS), and microbiological limits (absence of E. coli, Salmonella). This level of documentation is essential for brand protection and regulatory submissions.
- GMP compliance: Facilities adhering to current GMP ensure that raw material handling, testing, and packaging meet the hygiene and traceability standards expected by global supplement brands, reducing the risk of contamination or recalls.
- Particle size uniformity: Specifications such as ≥95% through 80 mesh facilitate homogeneous blending and consistent dosing, particularly in direct‑compression applications, thereby minimising batch‑to‑batch variation in final products.
From a compliance standpoint, while UMP is not currently classified as a Novel Food in the EU nor does it have an NDI notification in the US, its history of safe use in infant formula and dietary supplements is well documented. Reputable suppliers provide regulatory dossiers to support market‑specific notifications, a crucial service for brands planning international launches.
Formulation Strategies: Synergy, Dosage, and Practical Stability
An effective formulation requires more than simply adding UMP to a premix. For R&D teams, early consideration of dose, synergy, and stability can significantly accelerate development timelines and reduce costly reformulations. Based on clinical data and commercial experience, a daily dose of 300–600 mg of Uridine 5'-Monophosphate Disodium is appropriate for cognitive applications, mirroring the active range used in the Japanese trial (UMIN‑CTR ID: UMIN000045507). For sports nutrition concepts that leverage UMP's role in cellular energetics, it is important to recognise that human clinical data specifically for athletic performance are not yet available. Consequently, any positioning must remain anchored to general energy metabolism support—for example, "supports ATP production and cellular energy"—without implying direct ergogenic or cardioprotective outcomes. This distinction is not merely semantic; it is a regulatory necessity that protects brands from enforcement action.
Synergistic combinations can amplify the ingredient's benefits:
- With DHA and choline: Animal studies show that UMP, when co‑administered with DHA and CDP choline, increases brain phospholipid levels and enhances learning and memory beyond the effects of DHA alone. This triad supports the synthesis of synaptic membranes, making it ideal for premium cognitive stacks and enabling a "complete brain health" positioning.
- With magnesium or B‑vitamins: Magnesium acts as a cofactor in nucleotide metabolism, while folate supports methylation pathways; together with Vitamin B12 (Methylcobalamin), they may enhance UMP's role in cellular energy and neurotransmitter synthesis, offering opportunities for multifunctional formulations.
Stability considerations during manufacturing are often underestimated. UMP is hygroscopic and should be protected from moisture throughout processing. Practical measures include:
- Maintaining relative humidity below 45% during blending and encapsulation.
- Avoiding prolonged exposure to temperatures above 40 °C.
- Using desiccant packaging for bulk storage and finished product bottles.
Importantly, aqueous solutions of UMP should be prepared fresh and used promptly, as the molecule can degrade over time in liquid media. For gummies or liquid shots, microencapsulation or dry‑blending with stabilisers may be required to maintain potency—an investment that pays off in extended shelf‑life and consumer satisfaction.
Supply Chain Resilience and Total Cost of Ownership
In today's volatile raw material landscape, the true cost of an ingredient extends far beyond its per‑kilogram price. For brands seeking a reliable Uridine 5'-Monophosphate Disodium supplier, evaluating total cost of ownership means considering:
- Batch‑to‑batch consistency: A supplier with validated HPLC methods and stringent in‑process controls reduces the risk of formulation adjustments or rejected lots, saving both time and capital. This consistency directly protects your brand's reputation and avoids costly production delays.
- Security of supply: Multiple production lines or strategic stock holding can mitigate disruptions from raw material shortages or logistical delays, ensuring that your product launches and ongoing sales are not jeopardised.
- Regulatory support: Access to comprehensive technical dossiers, stability data, and toxicological summaries accelerates market entry and lowers the cost of compliance, particularly when entering multiple jurisdictions.
For a pure, GMP‑certified UMP powder, the ability to provide full traceability—from synthesis to finished product—is a hallmark of a professional partner. This includes not only the physical attributes but also the ethical sourcing of precursors and adherence to environmental standards. When evaluating distributors or exporters, request evidence of third‑party audits (e.g., FSSC 22000) and ask about their quality agreement process. Such diligence ensures that the bulk UMP you purchase will perform identically from the first kilo to the next, safeguarding your brand's equity and reducing hidden costs associated with variability.
Next Steps for Your Product Development
Based on the scientific evidence and practical insights outlined above, integrating Uridine 5'-Monophosphate Disodium into your product pipeline offers a scientifically grounded opportunity to differentiate in the cognitive health and sports nutrition categories. By partnering with a supplier that prioritises rigorous quality control, comprehensive documentation, and formulation support, you mitigate technical risks and accelerate time‑to‑market. To evaluate the suitability of this ingredient for your specific application, requesting a complimentary sample and the accompanying technical dossier is the logical next step. This package includes a batch-specific Certificate of Analysis, stability summary, and suggested formulation parameters—everything your R&D team needs to make an informed decision.
Frequently Asked Questions
Share this product
Related Products
Ready to get started?
Contact our team for technical specifications, pricing, and customized solutions.