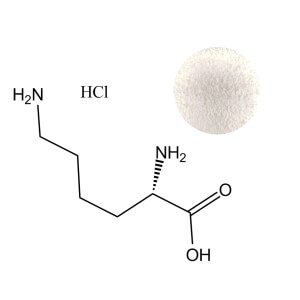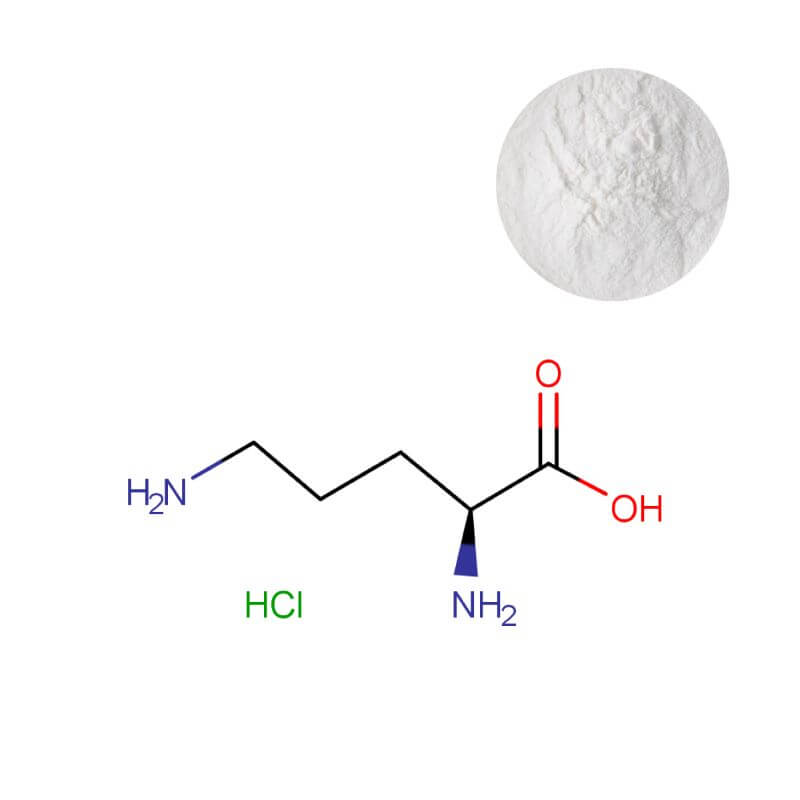
L-Ornithine HCl Powder
| Product Name | L-Ornithine HCl Powder |
| CAS Number | 3184-13-2 |
| Appearance | White crystalline powder |
| Purity | ≥ 99.0% (on dry basis) |
| Packaging | 1 kg/foil bag; 25 kg/cardboard drum |
| MOQ | 1 kg |
L-Ornithine HCl Powder (≥99.0%): A Clinically-Supported Amino Acid for Fatigue Management & Liver Support Formulations
For brands developing sports recovery blends, stress management formulas, or liver health products, standing out requires more than a standard amino acid matrix. The challenge lies in selecting ingredients with both a clear mechanism of action and robust safety data. L-Ornithine HCl powder, a non-protein amino acid central to the urea cycle, addresses this need. Premium material is typically produced via microbial fermentation, ensuring a 100% L-isomer, high solubility, and batch-to-batch consistency verified by HPLC. This makes it a strategic choice for formulations targeting fatigue reduction, ammonia clearance, and metabolic balance. In an increasingly crowded adaptogen and sports nutrition market, relying on ≥99.0% purity with published human data directly enables brands to build a defensible, science-backed claim, moving beyond generic amino acid blends and justifying premium pricing.
Clinical Evidence: How L-Ornithine HCl Supports Fatigue Reduction and Stress Response
L-Ornithine plays a critical role in converting toxic ammonia—generated during intense exercise or protein metabolism—into urea for excretion. But beyond this core biochemical function, recent human trials have quantified its direct benefits for mental and physical fatigue. A 2024 randomized, double-blind, placebo-controlled trial (doi: 10.3390/jcm13247583) administered 1,600 mg/day of L-ornithine for seven days to healthy volunteers exposed to the Trier Social Stress Test. Results showed significant improvements in the “fatigue-inertia” and “anger-hostility” dimensions of the Profile of Mood States (POMS), independent of cortisol changes. From a strategic formulation perspective, this suggests L-Ornithine HCl is not merely an exercise-recovery ingredient; it can also support stress-induced fatigue management, a growing segment in cognitive health and adaptogenic blends. For brand owners, this dual-application profile reduces the need to maintain separate raw material inventories for sports and mood support lines, directly lowering working capital requirements and accelerating time-to-market for multi-category product portfolios.
For procurement teams, this directly translates to a dual-application ingredient: one that serves both sports nutrition (post-workout recovery) and everyday wellness (stress-related tiredness). When sourcing bulk L-Ornithine HCl powder, look for suppliers who provide batch-specific COA confirming the ≥99.0% purity specification used in these clinical studies. This ensures your finished product’s efficacy aligns with published research, a key factor for regulatory submissions and consumer marketing.
Safety Benchmarks: What Defines a Premium, Trustworthy L-Ornithine HCl Ingredient?
While efficacy drives initial interest, safety data closes the deal for professional buyers. A 2025 systematic review (doi: 10.1007/s00726-025-03455-4) evaluated L-ornithine hydrochloride across 22 clinical trials, covering doses from 398 mg up to 14,025 mg per day and durations up to 156 days. The no-observed-adverse-effect level (NOAEL) was established at 12,000 mg/day, with gastrointestinal discomfort being the only notable event—and its risk difference was 0.00 compared to placebo. Key safety specifications for a premium ingredient include:
- Heavy metals: Lead ≤1.0 mg/kg, Arsenic ≤1.0 mg/kg, Cadmium ≤0.5 mg/kg, Mercury ≤0.1 mg/kg (ICP-MS method).
- Microbiological purity: Total plate count ≤1,000 CFU/g, absence of E. coli, Salmonella, and S. aureus.
- Residue on ignition: ≤0.10% and loss on drying ≤0.5% to ensure stability and low inorganic impurities.
From a compliance standpoint, a GMP-certified L-Ornithine HCl powder with full traceability and a certificate of analysis (COA) that meets USP standards is non-negotiable. For brands exporting to the EU or US, these specifications directly support your regulatory filing (e.g., NDI or Novel Food status). It’s worth noting that while lower-purity grades exist (e.g., 98%), premium formulations for human nutrition consistently rely on ≥99.0% material to minimize related substances and ensure predictable dissolution—a critical factor for capsule and stick-pack manufacturing. When viewed through a total cost of ownership (TCO) lens, the marginally higher upfront cost of ≥99.0% purity material prevents expensive production line stoppages due to inconsistent flow or dissolution failures, and eliminates the risk of finished product recalls caused by off-spec impurities – a liability that can easily exceed six figures.
Formulation in Practice: Stability, Synergy, and Dosage Guidelines
L-Ornithine HCl powder is highly water-soluble (pH 5.0–6.0 in 1% solution) and exhibits mild hygroscopicity. This makes it ideal for instant drink mixes, effervescent tablets, and two-piece capsules, but requires humidity control during production (recommended RH <45%). In solid dosage forms, the typical particle size (60–100 mesh) flows well but may need blending with anti-caking agents like silicon dioxide if used in high-humidity environments. For synergistic formulations, L-Ornithine pairs exceptionally well with:
| Co-ingredient | Synergy Mechanism | Typical Ratio (Orn:Partner) | Application |
|---|---|---|---|
| L-Arginine HCl | Both stimulate the urea cycle and growth hormone release; complementary ammonia scavenging. | 2:1 (Arg:Orn) | Post-exercise recovery, muscle protein synthesis. |
| L-Citrulline | Increases plasma ornithine and arginine levels; enhances nitric oxide production. | 1:1 or 2:1 (Cit:Orn) | Endurance and vascular health formulas. |
| Instantized BCAAs (2:1:1) | BCAAs reduce perceived fatigue, while ornithine clears ammonia – a two-pathway fatigue reduction. | 4:1 (BCAA:Orn) | Intra-workout or recovery RTD powders. |
Recommended daily dosages based on clinical research: for fatigue and stress management, 400–1,600 mg/day (Miyake 2014, 2024 RCT); for athletic performance support, 2,000–6,000 mg/day in divided doses. An effective formulation requires starting with a stability study: test your blend’s water activity (aim for Aw ≤0.30) and use moisture-barrier primary packaging (foil stick packs or desiccant-lined bottles). From a strategic formulation perspective, this prevents caking and preserves dissolution rate, directly impacting consumer acceptance and shelf-life claims. For tablet applications, direct compression is feasible, but pre-blending with a small amount of magnesium stearate (0.5–1%) improves flow without affecting dissolution.
Mitigating Risk: A Procurement Checklist for Sourcing Bulk L-Ornithine HCl Powder
For procurement teams, the price per kilogram is only one variable. The total cost of ownership includes quality consistency, supply chain reliability, and regulatory support. Below is a vendor evaluation framework used by industry experts:
- Documentation integrity: Does the supplier provide a batch-specific COA that includes all heavy metals (Pb, As, Cd, Hg), residual solvents (USP <467>), and microbiological tests? Are HPLC chromatograms available upon request?
- Production traceability: Is the L-Ornithine HCl produced via fermentation (preferred for non-GMO, vegan status) or chemical synthesis? Can they provide a flow chart from raw material to finished powder?
- GMP and third-party audits: Look for ISO 22000 or FSSC 22000 certification. Avoid suppliers who only claim “GMP compliant” without a verifiable certificate.
- Supply chain flexibility: Can they offer MOQ of 1 kg for pilot batches and scale up to 25 kg drums or metric tons without changing specifications?
- Regulatory support: Do they provide a technical dossier including stability data, allergen statement, and non-GMO declaration? This directly reduces your compliance risk when filing for NDI or Novel Food.
For procurement teams, this directly translates to lower risk of batch rejection, fewer production line stoppages due to inconsistent flow properties, and faster market access. A reliable supplier will offer a free sample (10–20 g) alongside a complete technical datasheet, allowing your R&D team to validate solubility and compatibility before committing to bulk orders. When you source bulk L-Ornithine HCl powder, prioritize partners who treat COA as a commitment, not a formality. A comprehensive due diligence process, including requesting stability data under accelerated conditions, can flag potential hygroscopicity issues before they trigger costly manufacturing delays – a classic TCO safeguard that experienced procurement leaders never overlook.
Next Steps: Accelerate Your Product Development with Verified L-Ornithine HCl
From clinical evidence on fatigue and stress to industry-grade safety benchmarks, L-Ornithine HCl powder offers a scientifically grounded, versatile amino acid for sports, liver, and cognitive health formulas. To de-risk your formulation project, request a complimentary sample and the complete technical dossier—including batch-specific COA, stability summary, and regulatory guidance. Request your sample and technical dossier today and validate how this premium ingredient performs in your specific matrix.
Frequently Asked Questions
Share this product
Related Products
Ready to get started?
Contact our team for technical specifications, pricing, and customized solutions.