Why Piperine 95% is a Must-Have Ingredient for Bioavailability
Bioavailability is the cornerstone of effective supplement formulation. This technical guide analyzes how Piperine 95% acts as a metabolic multiplier, offering R&D teams proven strategies to enhance nutrient absorption and product efficacy.
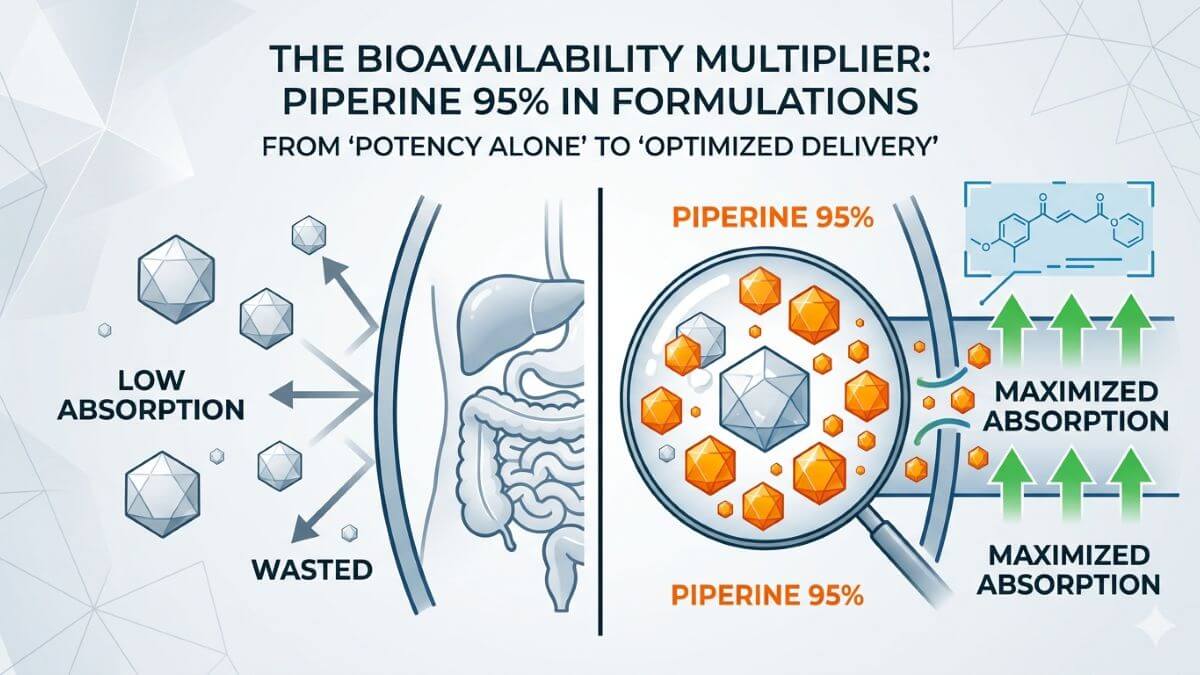
In the competitive landscape of functional supplements, the efficacy of a formula hinges on one critical factor: bioavailability. For years, brands have invested heavily in high-potency active ingredients, only to find that consumers experience minimal benefits because the body simply does not absorb them. This disconnect between ingredient promise and physiological reality has become a key pain point for R&D and procurement leaders. The solution lies not in adding more active compounds, but in strategically incorporating a proven bioavailability enhancer. This article explores the science behind bioavailability, the mechanism of piperine—the active compound in black pepper—and how it can serve as the strategic anchor for high-absorption formulations that deliver tangible results.
The Bioavailability Bottleneck: Why Potency Alone Is Not Enough
Many promising nutraceuticals—curcumin, coenzyme Q10, resveratrol—share a common limitation: poor oral bioavailability due to low aqueous solubility, rapid metabolism, or active efflux from enterocytes. Even when consumed in high doses, these compounds often fail to reach systemic circulation at therapeutically relevant levels. For brand owners, this translates into wasted formulation budgets, underwhelming consumer experiences, and ultimately, missed market opportunities. The key to overcoming this bottleneck lies in understanding the molecular gatekeepers that control absorption. What this means for procurement and R&D teams: investing in high-concentration actives without addressing absorption is akin to pouring water into a bucket with holes. The market has moved past the era of “more is better”—today’s successful formulas are built on efficiency, not excess.
Piperine 95%: The Molecular Key to Absorption
Piperine, the bioactive alkaloid derived from black pepper, has emerged as one of the most extensively studied bioavailability enhancers in the dietary supplement industry. Its mechanism extends far beyond simple solubilization. A comprehensive 2025 review published in Pharmaceutical Research (doi: 10.1007/s11095-025-03920-5) outlines how piperine modulates key metabolic pathways to improve drug and nutrient absorption. Specifically, it acts as a dual inhibitor of:
- Cytochrome P450 3A4 (CYP3A4): The primary phase I metabolic enzyme responsible for the oxidative metabolism of over 50% of drugs and many phytochemicals. Piperine reduces first-pass metabolism, allowing more of the active compound to reach systemic circulation.
- P-glycoprotein (P-gp): An efflux transporter that pumps xenobiotics back into the intestinal lumen. By inhibiting P-gp, piperine increases the net absorption of substrates like curcumin and coenzyme Q10.
Additionally, piperine enhances gut permeability and modulates phase II conjugation enzymes, further amplifying the bioavailability of co-administered nutrients. For R&D teams evaluating Black Pepper Extract (Piperine), these well-documented mechanisms provide a scientific foundation for formulating truly effective products. The takeaway for brands: piperine is not merely an additive—it functions as a “bioavailability multiplier” that can transform a standard formulation into a premium, results-driven offering without requiring reformulation of the active ingredients themselves.
Synergistic Pairings: Turning Challenging Actives into High-Performance Formulas
The true value of piperine lies in its ability to unlock the potential of other ingredients. Below are three clinically supported combinations that demonstrate its role as a bioavailability multiplier. Each pairing represents a proven shortcut to efficacy—allowing brands to bypass years of formulation trial and error.
1. Piperine + Curcumin: The Gold Standard Revisited
The classic pairing of curcumin with piperine has been validated by human clinical research. A landmark 1998 study (doi: 10.1055/s-2006-957450) demonstrated that co-administration of 2 g curcumin with 20 mg piperine increased the bioavailability of curcumin by a remarkable 2000% in human volunteers. This synergistic effect is achieved through piperine’s inhibition of intestinal glucuronidation, which would otherwise rapidly eliminate curcumin. While newer delivery systems continue to emerge, the piperine-curcumin duo remains a benchmark for cost-effective, science-backed formulation. Why this matters for brand strategy: launching a curcumin product without piperine today is increasingly seen as a substandard approach by discerning consumers and retailers. For brands developing anti-inflammatory or joint health products, sourcing standardized Curcumin Extract Powder with piperine ensures both efficacy and regulatory familiarity—while signaling to buyers that the formula is backed by foundational science.
2. Piperine + Coenzyme Q10: Enhancing Cardiovascular Support
Coenzyme Q10 (CoQ10) is a lipophilic molecule essential for mitochondrial energy production, yet its high molecular weight and poor water solubility severely limit oral absorption. A double-blind, randomized study published in the Journal of Nutritional Biochemistry showed that adding piperine to a CoQ10 formulation significantly increased the area under the plasma concentration-time curve (AUC) by 30%, compared to CoQ10 alone. The strategic implication: in the competitive cardiovascular health segment, where consumers are often price-sensitive, piperine offers a way to differentiate without increasing active ingredient dosage—preserving margin while delivering superior absorption data. For brands targeting this category, incorporating piperine with CoQ10 (Ubiquinone) Powder can be the decisive factor that moves a product from “generic” to “preferred.”
3. Piperine + Resveratrol: Improving Polyphenol Efficacy
Resveratrol, a potent polyphenol celebrated for its anti-aging and antioxidant properties, suffers from rapid glucuronidation and sulfation, resulting in extremely low systemic availability. Preclinical research has indicated that piperine can inhibit these conjugation pathways, leading to significantly higher plasma levels of free resveratrol. A study using mixed micelle formulations with piperine demonstrated a 5.7-fold increase in oral bioavailability compared to pure resveratrol. What this means for innovation-focused brands: while novel delivery technologies like liposomes capture headlines, piperine remains the most accessible and cost-effective tool for enhancing polyphenol absorption. When formulating anti-aging or metabolic health supplements, pairing piperine with Resveratrol Powder offers a straightforward strategy to boost efficacy without altering the core active ingredient—allowing brands to allocate R&D budgets to other differentiating factors.
Formulation Parameters: Selecting the Right Piperine 95%
Not all piperine extracts are created equal. For procurement and R&D teams, evaluating piperine suppliers requires attention to several critical quality attributes. The following table outlines key specifications that directly impact bioavailability outcomes. The underlying insight: in B2B sourcing, the cheapest piperine is rarely the most cost-effective when inconsistent purity or poor particle size leads to batch variability and compromised product performance.
| Parameter | Specification Range | Impact on Bioavailability |
|---|---|---|
| Piperine Content | ≥95.0% by HPLC | Ensures consistent potency; lower purity may yield variable absorption results. |
| Particle Size | D90 ≤ 100 μm | Fine particle size enhances dissolution and rapid release in the GI tract. |
| Solvent Residues | ≤ 50 ppm (ethyl acetate, ethanol) | Meets GMP and food-grade safety standards; critical for clean-label claims. |
| Heavy Metals | ≤ 1 ppm (lead, arsenic) | Essential for compliance with international regulations (e.g., Prop 65). |
| Microbial Limits | < 1000 CFU/g total plate count | Guarantees stability and safety throughout shelf life. |
Key Formulation Considerations for R&D Teams
Incorporating piperine into a formula requires attention to dosage, compatibility, and manufacturing constraints. Below is a checklist of formulation parameters that ensure optimal performance. What experienced formulators know: piperine’s versatility is its greatest asset—it integrates seamlessly into most manufacturing workflows, making it one of the lowest-risk efficacy boosters available.
- Effective Dosage: Clinical studies typically use 5–20 mg of piperine per serving. A 10 mg dose is common for most synergist applications. Implication: low-dose requirement means minimal impact on final product cost.
- Stability: Piperine is stable under standard dry granulation and encapsulation conditions. For liquid formulations, pH should be maintained between 4.0 and 7.0 to prevent degradation. Implication: works across nearly all delivery formats without special handling.
- Synergist-to-Active Ratio: Ratios vary by active ingredient. For curcumin, a 1:100 ratio (20 mg piperine to 2 g curcumin) is classic; for CoQ10, 5–10 mg piperine with 100–200 mg CoQ10 is typical. Implication: flexibility allows customization based on brand positioning and cost targets.
- Clean Label Compatibility: Piperine is naturally derived and can be listed as “black pepper extract” or “piperine,” aligning with consumer preferences for familiar ingredients. Implication: supports premium positioning without requiring complex labeling narratives.
Safety and Regulatory Perspective
Piperine has been recognized as safe (GRAS) by the U.S. FDA for use in food and supplements at levels consistent with good manufacturing practices. The European Food Safety Authority (EFSA) has established an acceptable daily intake of up to 5 mg/kg body weight, which translates to a wide safety margin for typical supplement doses (5–20 mg/day). However, formulators should be aware that piperine’s enzyme-inhibiting properties may interact with certain prescription medications, particularly those metabolized by CYP3A4 (e.g., statins, calcium channel blockers). Responsible suppliers provide comprehensive documentation, including Certificates of Analysis (COA) and GMP compliance, to support safe and compliant product development. The regulatory bottom line for procurement: selecting a supplier that offers full traceability and batch-specific COAs is not a “nice-to-have”—it is the foundation of defensible product claims and long-term market access.
Conclusion: Elevating Formulations with a Strategic Bioavailability Anchor
In an era where consumers demand measurable results, the difference between a “me-too” product and a market leader often comes down to bioavailability. Piperine 95% offers a scientifically validated, cost-effective, and versatile solution to unlock the full potential of otherwise challenging active ingredients. By strategically incorporating piperine into formulas featuring curcumin, CoQ10, resveratrol, and other poorly absorbed compounds, brands can deliver superior efficacy, strengthen their scientific storytelling, and build lasting consumer trust. The final takeaway: bioavailability is not a secondary consideration—it is the foundation upon which credible supplement brands are built. For a deeper dive into technical specifications, stability data, and custom formulation support, explore the detailed product pages or contact our technical team for consultation.
Share this article
Found this helpful? Share it with others!
Related Products
Products mentioned in this article
Want to learn more?
Explore our products or contact our team for personalized solutions and expert advice.