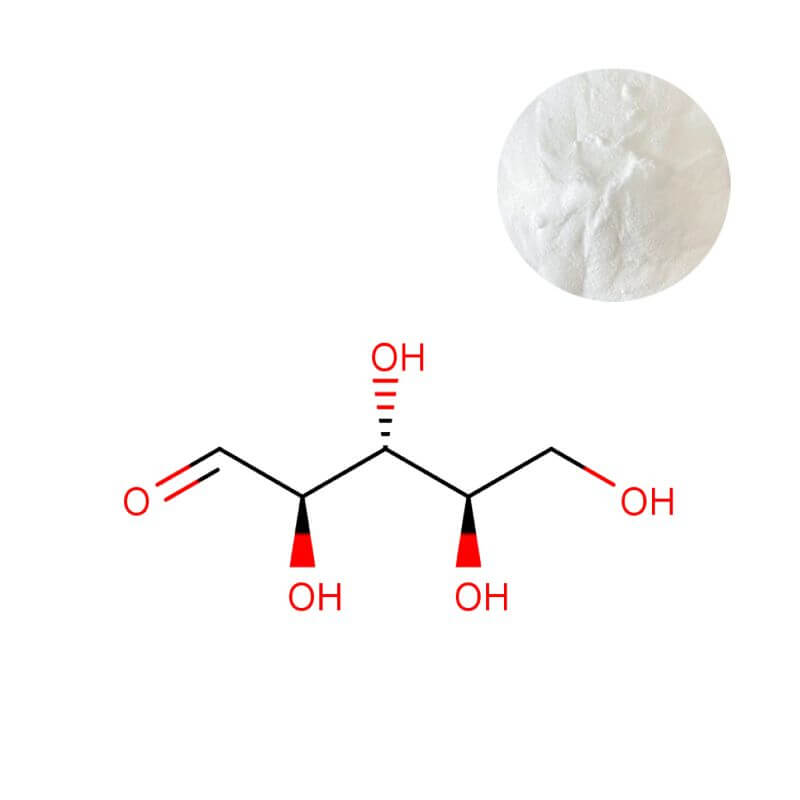
D-Ribose Powder
| Product Name | D-Ribose Powder |
| CAS Number | 50-69-1 |
| Appearance | White crystalline powder |
| Purity | 99.0% min. (by HPLC) |
| Packaging | 1 kg/bag, 5 kg/bag, 25 kg/drum |
| MOQ | 1 kg |
D-Ribose Powder: Sustained Cellular Energy for Premium Sports & Cardiovascular Formulations
When developing advanced nutraceuticals, the difference between a generic product and a market leader often lies in the depth of its foundational ingredients. For brands aiming to differentiate in the crowded sports nutrition and cardiovascular health categories, selecting a clinically validated ingredient with a stable supply chain is the critical first step. D-Ribose has emerged as a non-negotiable component for formulators addressing energy decline, muscle recovery, and cardiac wellness. This naturally occurring pentose sugar is the structural backbone of ATP, the cell's primary energy currency. However, not all D-Ribose powders deliver the same consistency. Premium material, characterised by ≥ 99.0 % purity (HPLC), a tight particle size distribution (20-80 mesh), and documented optical rotation, ensures batch-to-batch reproducibility. For brands sourcing bulk D-Ribose, understanding these specifications is key to protecting your product's reputation. This article explores how to leverage this essential ingredient effectively—from its science-backed mechanisms to the sourcing and formulation decisions that accelerate regulatory success.
The Science Behind D-Ribose: How It Fuels ATP Resynthesis and Supports Cardiac & Muscle Function
D-Ribose is not merely a sweetener; it is the rate-limiting substrate for the salvage pathway that rebuilds ATP pools after intense metabolic demand. In cardiomyocytes and skeletal muscle, energy depletion during ischaemia or exhaustive exercise triggers a loss of adenine nucleotides. Exogenous D-Ribose, once phosphorylated to ribose-5-phosphate, drives the pentose phosphate pathway and supplies 5-phosphoribosyl-1-pyrophosphate (PRPP)—the essential molecule for de novo purine synthesis and nucleotide salvage. This mechanism directly accelerates the recovery of ATP, enabling tissues to regain function faster.
A robust randomised controlled trial published in The American Journal of Cardiology provides quantifiable clinical support. In a 12-week study of 216 patients with heart failure with preserved ejection fraction (HFpEF), supplementation with 15 g/day of D-Ribose significantly improved:
- Kansas City Cardiomyopathy Questionnaire clinical summary score (improvement of 17.3–25.8 points).
- Vitality score (7.7–8.2 points).
- Ejection fraction (increase of 7.1–8.0 %).
- B-type natriuretic peptide and lactate/ATP ratio, both markers of cardiac stress (doi: 10.1016/j.amjcard.2022.04.031).
What does this mean for your product portfolio? It confirms that D-Ribose can underpin credible structure/function claims for cardiovascular health, sports recovery, and healthy ageing. By including it in formulations targeting active individuals or aging populations, brands can address the core consumer demand for measurable vitality rather than temporary stimulation. For R&D teams, these clinical endpoints provide a solid foundation for label claims that resonate with informed consumers, building trust in a competitive landscape.
Quality Benchmarks: What to Look for in a Premium D-Ribose Ingredient
For B2B buyers, the technical dossier is as important as the ingredient itself. A reliable D-Ribose supply is defined by strict adherence to internationally recognised specifications. Beyond a minimum purity of 99.0 % (HPLC), a high-quality material will exhibit a consistent specific rotation between –18.0° and –22.0° (c=2, H₂O), confirming the correct stereochemistry essential for bioactivity. Particle size uniformity (20–80 mesh) ensures predictable flow and blend homogeneity, directly impacting tableting or encapsulation efficiency. From a total cost of ownership perspective, a slightly higher-priced material that guarantees batch-to-batch consistency can eliminate expensive rework and compliance delays—factors often overlooked by procurement focused solely on upfront cost. When evaluating a D-Ribose supplier, these quality indicators should be non-negotiable.
From a regulatory standpoint, the European Food Safety Authority (EFSA) has evaluated D-Ribose as a novel food and concluded that it is safe under the proposed conditions of use. The EFSA statement establishes a tolerable upper intake of 36 mg/kg body weight/day, based on a NOAEL of 3.6 g/kg bw/day from subchronic studies (doi: 10.2903/j.efsa.2018.5485). For brands targeting EU markets, sourcing from a partner who provides batch-specific Certificates of Analysis (COA) with full heavy metal (Pb ≤ 0.5 mg/kg, As ≤ 0.5 mg/kg, Cd ≤ 0.5 mg/kg, Hg ≤ 0.1 mg/kg) and microbiological data (TPC ≤ 1,000 CFU/g, pathogens absent) is not optional—it is the foundation for a compliant finished product. Experienced quality assurance teams routinely audit these data points to avoid costly regulatory surprises when entering new markets.
Practically speaking, a comprehensive COA should also include residual solvent analysis (USP <467>) and loss on drying (≤ 0.5 %). These data points reassure quality assurance teams that the ingredient will remain stable throughout its 24-month shelf life when stored in tight, light-resistant containers.
Formulation in Practice: Stability, Synergy, and Delivery Options
Incorporating D-Ribose into finished products requires attention to its physicochemical behaviour. As a highly water-soluble sugar (approximately 100 g/100 mL at 20 °C), it dissolves readily in beverage matrices, but its hygroscopic nature demands controlled manufacturing environments. Maintaining relative humidity below 45 % during blending and encapsulation prevents clumping and ensures uniform fill weights. Common dosage forms and corresponding considerations are outlined below. Early consultation with your ingredient supplier on the optimal particle size can significantly reduce scale-up risks and accelerate time-to-market.
| Dosage Form | Typical D-Ribose Inclusion | Formulation Considerations |
|---|---|---|
| Powder blends / stick packs | 1.5–5 g per serving | Use free-flowing agglomerated grade to reduce dust and improve dissolution. |
| Hard capsules | 500 mg – 1 g per capsule | Blend with lubricants (e.g., magnesium stearate) to improve flow; particle size 40–80 mesh recommended. |
| Tablets | 250 mg – 1 g per tablet | Direct compression possible; avoid over-lubrication to maintain hardness. |
| Ready-to-drink beverages | Up to 5 g per bottle | Stable at pH 3–7; pasteurisation does not degrade D-Ribose. |
Synergy with other ingredients amplifies the value proposition. D-Ribose pairs exceptionally well with:
- Dimagnesium Malate: a cofactor in ATP synthesis.
- CoQ10 Ubiquinone: supports mitochondrial electron transport, complementing D-Ribose's role in ATP production.
- L-Carnitine L-Tartrate: facilitates fatty acid oxidation, providing an alternative energy source.
For brands developing a comprehensive energy or heart health stack, a combination of D-Ribose (5–10 g), Coenzyme Q10 (100–200 mg), and magnesium (100–200 mg) offers a multi-pathway approach. This not only enhances efficacy but also creates a unique selling proposition that distinguishes a product from single-ingredient offerings. Such proprietary blends can be protected through formulation patents, adding long-term brand value.
Market Trends: Positioning D-Ribose for Sports, Heart, and Healthy Aging
Consumer interest in "cellular health" is accelerating. Today's informed buyers look beyond superficial energy boosts and seek ingredients that support mitochondrial function—the core of vitality. D-Ribose fits seamlessly into three high-growth categories:
- Sports nutrition: marketed as a recovery accelerator that reduces delayed-onset muscle soreness and replenishes energy stores post-workout.
- Cardiovascular health: positioned for aging consumers who wish to maintain an active lifestyle and support heart muscle energy.
- Healthy aging / energy support: incorporated into daily wellness powders targeting fatigue and metabolic decline.
An emerging product concept is the "mitochondrial health blend" combining D-Ribose with NAD+ precursors (e.g., nicotinamide riboside) and adaptogens. Such a formulation speaks directly to consumers seeking sustained energy without caffeine-like crashes. For brands aiming at the premium longevity segment, this combination offers a science-forward positioning that justifies higher price points. From a formulation cost perspective, using a high-purity D-Ribose (≥ 99.0 %) sourced from a reliable D-Ribose supplier ensures that the active component performs predictably, allowing the brand to focus on marketing innovation rather than troubleshooting quality issues.
Next Steps for Your Product Development
Integrating D-Ribose into your pipeline is a strategic move toward building credible, science-backed products. The key to success lies in partnering with a D-Ribose supplier that provides full transparency—batch-specific COAs, regulatory documentation (including EFSA novel food status), and technical support for formulation challenges. By evaluating a sample and the complete technical dossier in your own lab, you can validate the ingredient's performance in your specific matrix, de-risk your development, and shorten your path to market.
Request your complimentary sample and technical dossier today to experience how a premium D-Ribose ingredient can elevate your next formulation.
Frequently Asked Questions
Share this product
Related Products
Ready to get started?
Contact our team for technical specifications, pricing, and customized solutions.