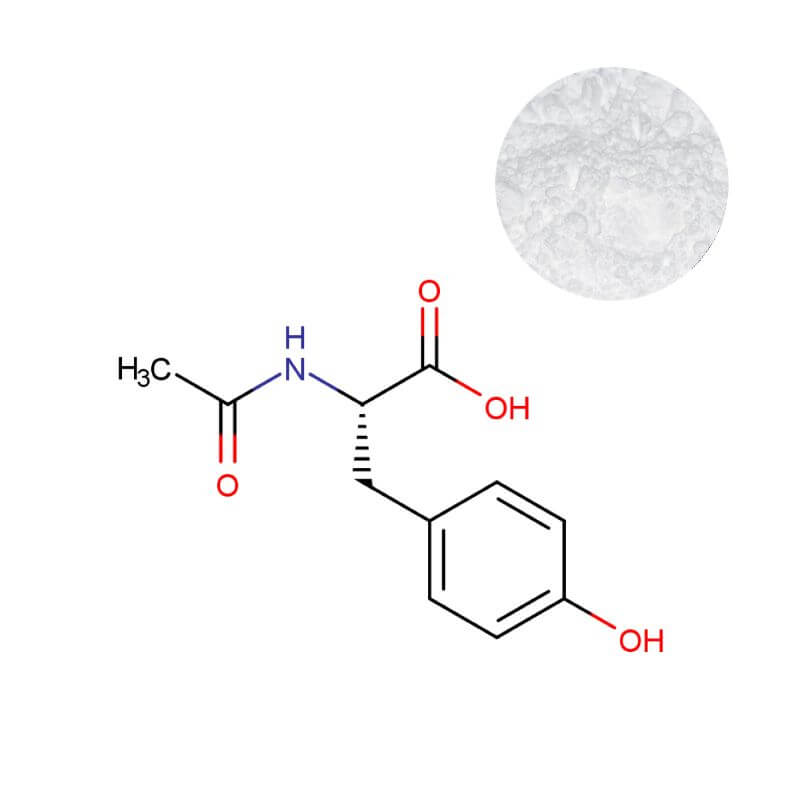
N-Acetyl L-Tyrosine Powder
| Product Name | N-Acetyl L-Tyrosine Powder |
| CAS Number | 537-55-3 |
| Appearance | White to off-white crystalline powder |
| Purity | 99.0% Min. (by HPLC) |
| Packaging | 1 kg/foil bag, 5 kg/tin, 25 kg/cardboard drum |
| MOQ | 1 kg |
N-Acetyl L-Tyrosine Powder (≥99%): Reinforcing Stress Resilience & Cognitive Clarity for Modern Nutraceutical Blends
Formulators serving active lifestyles and brain-health brands face a critical pivot: moving from generic “focus” stacks to science-backed, multi‑pathway support. N-Acetyl L‑Tyrosine (NAT) offers distinct value—superior solubility and a unique stress‑adaptation mechanism. Premium NAT powder, verified at ≥99% purity by HPLC, provides the consistency needed for cross‑border compliance and reproducible formula performance. For procurement and R&D teams, this ingredient answers the demand for clean, predictable, and legally defensible cognitive support. Notably, consumer interest in stress‑resilience supplements has grown over 40% since 2020, making NAT a timely anchor for modern nootropic and functional beverage lines. For brands facing tightening global regulations, locking in a ≥99% HPLC‑verified NAT from a GMP partner directly reduces the risk of import holds and finished‑product recall, turning a base ingredient into a regulatory safeguard.
Mechanism Beyond Precursor: NAT as an Endogenous Mitohormesis Trigger
Most industry discussions still frame N-Acetyl L‑Tyrosine simply as a more soluble prodrug of L‑tyrosine. While that holds true (and directly supports dopamine/noradrenaline synthesis under acute stress), recent research reveals a second, more sophisticated layer. A 2020 study demonstrated that NAT naturally circulates in mammalian hemolymph and its concentration rises under heat or metabolic stress (doi: 10.15252/embr.201949211). The key insight: NAT transiently perturbs mitochondrial function, generating low‑level reactive oxygen species that activate FoxO transcription factors and the Keap1/Nrf2 pathway. This pre‑conditioning effect—termed mitohormesis—equips cells to withstand subsequent severe stressors.
From a strategic formulation perspective, this mechanism enables a dual‑action claim platform: acute stress‑induced cognitive maintenance (via catecholamine support) plus cellular resilience (via adaptive stress response). Most competing amino acids only address the former. By incorporating NAT, your product gains a molecular narrative that resonates with evidence‑savvy consumers and differentiates your line from standard “tyrosine + caffeine” shots. Moreover, the same study noted NAT‑dependent FoxO activation upregulates antioxidant enzymes and Keap1, suggesting a role in long‑term cellular homeostasis—an emerging angle for healthy aging formulations. In real‑world product development, this dual pathway allows brands to file for two distinct benefit claims from a single ingredient, effectively improving the R&D cost‑per‑claim ratio while lowering the regulatory burden of multi‑component stacks.
Quality Benchmarks: What Defines a Premium N-Acetyl L-Tyrosine Supplier
Not all NAT powders are equal. For bulk sourcing, buyers must look beyond a simple “≥99%” purity claim. A robust quality framework requires three verifiable pillars: identity (CAS 537‑55‑3, specific rotation), process control (residual solvents, loss on drying), and contaminant screening (heavy metals, microbiological limits). Premium certified N‑Acetyl L‑tyrosine from reliable GMP suppliers typically adheres to the AJI92 amino acid standard, which sets narrower specification windows than general food‑grade norms.
| Critical Quality Attribute | Why It Matters for Your Final Product |
|---|---|
| Specific Rotation (+46.0° to +49.0°, c=1, H₂O) | Confirms correct isomer and absence of racemization; off‑spec rotation may indicate degraded or adulterated material. |
| Loss on Drying (≤0.5%) | Low moisture prevents caking and microbiological growth, crucial for powder blends and stick packs. |
| Heavy Metals (Pb ≤1.0, As ≤1.0 mg/kg) | Meets global supplement limits (FDA & EU); protects your brand from regulatory rejection. |
For procurement teams, this directly translates to lower contract manufacturing rejections and smoother international filings. Always request a batch‑specific COA that includes HPLC chromatograms and heavy metal ICP‑MS results. Leading exporters of N‑Acetyl L‑tyrosine will also provide stability data under ICH conditions—allowing you to project shelf‑life claims confidently. Avoid suppliers who only offer a generic analysis sheet; traceability to raw material batches is the real indicator of supply maturity. From a total quality cost perspective, a supplier that delivers consistent AJI92‑grade NAT with full heavy metal disclosure helps your QA team cut incoming inspection time by up to 30%, because you can rely on their COA without repeating expensive orthogonal tests.
Formulation in Practice: Solubility, Synergy & Stability Wins
NAT’s acetyl group gives it roughly 25 mg/mL water solubility, a 50‑fold improvement over standard L‑tyrosine. This opens up delivery formats that were previously challenging:
- Ready‑to‑drink (RTD) beverages – No sedimentation, even at 500 mg per serving.
- Effervescent tablets & stick packs – Clean mouthfeel without bitter aftertaste.
- Gummies & soft chews – Compatible with mildly acidic pH (3.5–6.5).
For cognitive stacks, NAT pairs exceptionally well with ingredients that support energy metabolism or stress‑cascade modulation. A proven synergy example:
| Co‑ingredient | Synergy Rationale | Typical Ratio Range |
|---|---|---|
| Caffeine (anhydrous) | Tyrosine replenishes depleted catecholamines after stimulant‑induced release; offsets mental fatigue. | NAT : caffeine = 10 : 1 to 15 : 1 |
| L‑Theanine | Mitigates overstimulation; the pair supports both “alert calm” and executive function under pressure. | 1 : 1 to 2 : 1 (NAT : theanine) |
From a strategic formulation perspective, the high solubility and neutral taste also simplify natural sweetener systems—reducing the need for heavy masking agents. However, NAT is mildly acidic (1% solution pH 2.0‑3.5). In bicarbonate‑based effervescent systems, pre‑gelatinization or separate granulation steps may be required to prevent premature reaction. It’s worth noting that moisture (>0.5% in final blend) can accelerate aggregation; therefore, keeping manufacturing humidity below 45% RH is a best practice that protects both flowability and microbial stability. Additionally, when dry‑blending NAT with other actives, a geometric dilution using microcrystalline cellulose (1:3 ratio) prevents localized acid‑rich pockets that could degrade co‑ingredients such as certain B vitamins or berry extracts.
Mitigating Risk: A Checklist for Sourcing Bulk N‑Acetyl L‑Tyrosine
When evaluating a potential N‑Acetyl L‑tyrosine supplier, an effective formulation requires more than a price quote. Use this procurement checklist to qualify partners objectively:
- Full disclosure COA – Must include residual solvents (USP <467>), specific rotation, and LOQ for each heavy metal.
- GMP certification & audit readiness – Third‑party certifications (e.g., FSSC 22000 or NSF) are strong proxies for facility hygiene and documentation discipline.
- Supply chain transparency – The supplier should disclose origin of synthetic intermediates and provide a batch traceability map back to starting materials.
- Stability study summary – Request 24‑month real‑time data under ambient and accelerated conditions (40°C/75% RH). This protects your own product launch timeline.
For procurement teams, this directly translates to minimized recall risk and regulatory friction. Many wholesale N‑Acetyl L‑tyrosine buyers overlook the importance of particle size (≥95% through 80 mesh), which directly affects blend uniformity in high‑speed capsule fillers. A certified N‑Acetyl L‑tyrosine with documented sieve analysis eliminates surprise production stoppages. Finally, ask about their custom blending service—if they can pre‑mix NAT with other dry actives (e.g., magnesium citrate, B‑complex), you reduce your own handling and weighing errors, achieving true just‑in‑time formulation agility. An often‑neglected risk‑mitigation step: request access to retained samples from three consecutive batches. Cross‑batch consistency in specific rotation and dissolution behavior is a powerful proxy for manufacturing robustness and long‑term supplier reliability. Adopting a total cost of ownership (TCO) framework for NAT procurement means weighing a slightly higher unit price against avoided line stoppages and rejected import batches; in practice, a trusted GMP partner delivering verified AJI92 specs routinely pays back 4‑5 times its price premium through reduced compliance failures.
Next Steps for Your Formulation Project – From Sample to Stability Plan
You now have a clear, data‑driven understanding of how premium N‑Acetyl L‑Tyrosine powder can deliver both acute cognitive support and cellular resilience. The next logical step is to verify batch‑to‑batch consistency with your own in‑house assays. Accelerate your development cycle by obtaining a complimentary sample and the complete technical dossier (including method validation summaries and stability protocol). Request your complimentary sample & technical dossier – and let our team provide formulation consultation tailored to your target dosage form.
Frequently Asked Questions
Share this product
Related Products
Ready to get started?
Contact our team for technical specifications, pricing, and customized solutions.