The Evolution of NAD+ Market in 2026: Why NR-Cl is the Strategic Compliant Choice for Global Brands?
The NAD+ market is maturing. This article decodes why NR-Cl offers ultimate compliance certainty for 2026, backed by head-to-head clinical evidence and PQQ synergy. A must-read roadmap for global brand R&D and procurement leaders.
The global market for NAD+ precursors is at a pivotal juncture. Once dominated by hype and speculative ingredient sourcing, the industry is rapidly maturing. Euromonitor data indicates the longevity sector is transitioning from a niche scientific area to a core health pillar, with consumers demanding science-backed solutions that target cellular health. However, alongside this growth comes increased regulatory scrutiny. For brands eyeing global expansion, the choice of which NAD+ booster to stake their portfolio on has never been more critical—or more complex. This article moves beyond surface-level trends to decode the strategic implications of recent regulatory shifts, scientific breakthroughs, and formulation science, offering R&D and procurement leaders a roadmap to future-proof their product decisions.
The Shifting Regulatory Landscape of NAD+ Precursors
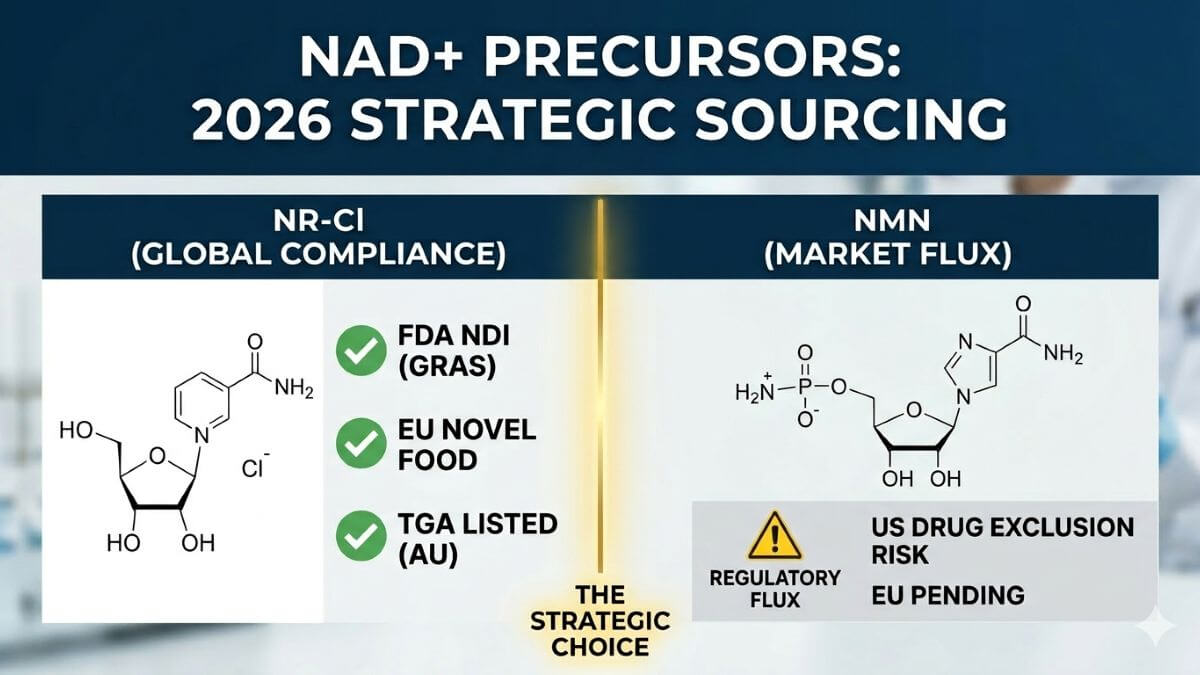
The past 18 months have reshaped the compliance map for NAD+ boosters. While both NR and NMN have established safety profiles, their regulatory trajectories now diverge significantly. For global brands, this divergence translates directly into supply chain reliability and market access timelines. The following table illustrates the current regulatory standing of each precursor in major markets:
| Regulatory Parameter | Nicotinamide Riboside Chloride (NR-Cl) | β-Nicotinamide Mononucleotide (NMN) |
|---|---|---|
| FDA GRAS Status | GRAS affirmed (multiple notifications) | Self-affirmed GRAS only; no FDA response letter |
| FDA NDI Notification | NDI approved (e.g., NDI 1134) | Kingdomway received NDI approval (Jan 2026); other suppliers pending |
| EU Novel Food | Authorised (Commission Implementing Regulation (EU) 2019/506) | Application under review; no approval yet |
| Regulatory History | Stable since 2013 GRAS notification | 2022 FDA attempted to exclude from supplement definition → 2025 reversal; ongoing uncertainty for non-NDI holders |
This table illustrates a clear pattern: NR-Cl enjoys established, uncontested status in all major markets, while NMN remains in flux despite its recent NDI approval for a single supplier. For brands targeting global distribution, relying on an ingredient with fragmented regulatory acceptance poses significant inventory and labeling risks. What this means for procurement teams: Choosing NR-Cl today means one formulation can serve North America, Europe, and Asia simultaneously, slashing time‑to‑market and regulatory duplication costs. In contrast, NMN still requires market‑by‑market validation, a luxury few emerging brands can afford. Yet compliance is merely the entry ticket. The real differentiator lies in the depth of clinical substantiation—and this is where the 2026 data delivers decisive clarity.
Head-to-Head Clinical Evidence: What the 2026 Data Shows
Regulatory compliance alone is insufficient; robust clinical substantiation drives consumer trust. Two recent studies provide pivotal insights.
A landmark randomized controlled trial published in Nature Metabolism directly compared three NAD+ boosters in healthy adults (doi: 10.1038/s42255-025-01421-8). After 14 days of supplementation, both NR and NMN equally elevated circulating NAD+ levels, while niacinamide produced only transient changes. Importantly, the study revealed that both NR and NMN interact with the gut microbiome to generate nicotinic acid, enhancing microbial metabolism—a novel mechanistic insight that strengthens the scientific foundation of both molecules. The implication for brands: NR is scientifically on par with NMN in boosting NAD+, eliminating any efficacy‑based reason to favour the more regulatorily uncertain ingredient. The microbiome connection also opens new storytelling angles about gut–brain or gut–energy axes.
Another rigorous 24-week trial focused on high‑dose NR (2000 mg/day) in individuals with long‑COVID symptoms (doi: 10.1016/j.eclinm.2025.103633). Published in eClinicalMedicine (The Lancet), the study reported a 2.6‑ to 3.1‑fold increase in NAD+ levels sustained for 20 weeks. While the primary cognitive endpoint was not met, exploratory analyses showed significant improvements in executive function, fatigue severity, sleep quality, and depressive symptoms after 10 weeks. These findings underscore NR's bioavailability and safety at high doses, supporting its use in targeted recovery formulations. What this means for product developers: NR's safety at 2 g per day—four times the typical dose—provides a wide therapeutic window. Brands can confidently explore high‑potency formulations or combine NR with other actives without fearing adverse interactions. The long‑COVID application also hints at broader opportunities in post‑viral recovery and stress management, markets that are rapidly expanding.
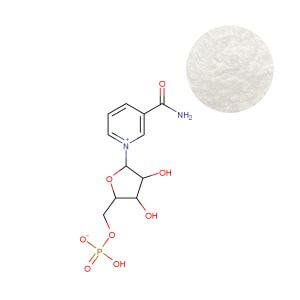
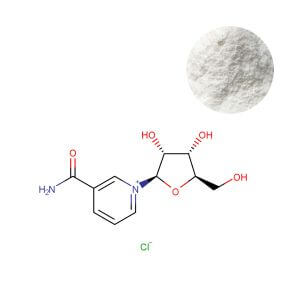
For procurement professionals, the key takeaway is that NR possesses clinical data equal to or exceeding that of NMN, with the added advantage of a longer, unbroken safety record in human trials. A stable, compliant ingredient with deep clinical roots, such as Nicotinamide Riboside Chloride Powder, provides the foundation for a sustainable product line. In contrast, while NMN Powder continues to generate consumer interest, its regulatory and scientific uncertainties make it a higher-stakes option for global portfolios.
Why NR-Cl Dominates the Formulation Landscape
Beyond compliance and clinicals, practical formulation attributes determine a brand's ability to innovate. NR-Cl (the chloride salt) offers distinct advantages:
- Stability: Crystalline form with demonstrated stability at 25 °C/60% RH for 24 months (loss ≤2.0% by HPLC). This translates to longer shelf‑life and fewer returns—critical for e‑commerce models where products may sit in warehouses for months.
- Purity: Available at ≥99.0% (HPLC) with full COA traceability. For brands, high purity means no need to overage to compensate for degradation, keeping formula costs predictable.
- Solubility: Freely soluble in water (>500 mg/mL), enabling versatile dosage forms. Whether developing ready‑to‑drink shots or gummies, formulators can work with NR‑Cl without encountering grittiness or precipitation.
- Regulatory Dossier: Complete technical packages for GRAS, NDI, and Novel Food are readily available, with suppliers routinely providing these with every shipment, speeding up your own regulatory filings and reducing legal consultation costs.
But even the best single ingredient can be elevated when paired intelligently—which brings us to the emerging trend of mitochondrial stacks.
The Synergistic Opportunity: Pairing NR with PQQ
To maximize product differentiation, leading brands are combining NR with complementary actives. PQQ Disodium Salt has emerged as the ideal partner because it targets mitochondrial biogenesis—a distinct but related pathway to NAD+ restoration.
- Purity: ≥99.0% by HPLC (disodium salt form).
- Dosing: Clinical studies typically use 20 mg/day.
- Mechanism: PQQ activates PGC‑1α, stimulating new mitochondria formation; NR replenishes NAD+ to fuel those mitochondria.
- Regulatory: PQQ is self-affirmed GRAS in the US and approved as a novel food in the EU and Japan.
This dual‑action "mitochondrial vitality" concept is already appearing in premium supplements and functional beverages. A 2024 review confirmed that PQQ (20 mg/day) for 8 weeks reduces subjective fatigue and improves sleep quality—benefits that align perfectly with NR's effects on energy and cognition. Combining them allows brands to tell a more compelling cellular health story. For R&D teams, this synergy means you can offer a solution that addresses both the quantity (biogenesis) and the fuel (NAD+) of mitochondria, a narrative that resonates deeply with biohackers and aging consumers. Moreover, by sourcing both ingredients from a single trusted supplier, you simplify procurement and ensure compatibility in your final product.
With the science and synergy in place, the final step is translating knowledge into a procurement strategy that secures your brand's future.
Strategic Sourcing Checklist for 2026
When evaluating NAD+ precursor suppliers, procurement teams should prioritize the following criteria:
- Regulatory Documentation: Request GRAS affirmation letters, NDI notifications, and EU Novel Food authorisation for the exact product you intend to buy. NR‑Cl suppliers routinely provide these; for NMN, only a handful can show a recent NDI.
- Traceability: Full chain of custody from synthesis to finished product, with batch‑specific COAs including residual solvent and heavy metal analysis. This protects your brand from contamination scandals and ensures every batch meets label claims.
- Stability Data: Accelerated and real‑time stability studies supporting the claimed shelf life under recommended storage conditions. Ask for data in the exact packaging you plan to use—this can prevent costly reformulations later.
- GMP Compliance: Third‑party audits (e.g., NSF, SGS) of manufacturing facilities. In an era of increasing FDA inspections of foreign facilities, GMP certification is non‑negotiable.
- Supply Capacity: Minimum annual production capacity and inventory buffer to ensure uninterrupted supply. The NAD+ category is growing rapidly; you don't want to be left out of stock during a demand surge.
NR-Cl, with its mature supply base and transparent regulatory history, consistently meets these criteria. Suppliers offering comprehensive dossiers enable brands to go to market faster and with lower legal risk. What this means for your bottom line: A well‑sourced NR‑Cl program can reduce regulatory delays by 6‑12 months and avoid the costly reformulations that plague brands tied to ingredients with shifting legal status.
Conclusion: Future‑Proof Your NAD+ Portfolio with NR-Cl
The NAD+ market is maturing from a speculative frontier to a regulated category. Brands that anchor their products on Nicotinamide Riboside Chloride gain immediate access to all major markets, backed by clinical data that withstands regulatory scrutiny. Whether positioned as a standalone ingredient or combined with mitochondrial supporters like PQQ, NR-Cl offers the compliance certainty that forward‑thinking brands require. The choice is no longer about which precursor is trendier—it's about which one lets you sleep soundly while your product ships globally.
Ready to explore technical specifications or request a sample? Contact our technical team to discuss how our premium NR-Cl and PQQ can accelerate your next product launch.
Share this article
Found this helpful? Share it with others!
Related Products
Products mentioned in this article
Want to learn more?
Explore our products or contact our team for personalized solutions and expert advice.