Beyond Basic B1: How Thiamine Form Innovation is Reshaping the Nutritional Marketplace
In a market shaped by supply volatility and proven efficacy demands, selecting the correct thiamine form—Benfotiamine, Mononitrate, or HCl—is key. This guide gives B2B leaders a framework to leverage these differences for product differentiation, supply chain resilience, and premium positioning.
The Commodity Trap: Why Price-Only Sourcing Is a Strategic Vulnerability
In the legacy procurement model, vitamin B1 was treated as a cost-per-kilogram line item. This approach is now dangerously obsolete. It exposes your operation to a dual threat: immediate supply chain disruption and long-term brand irrelevance. The 2024-25 Thiamine Mononitrate market is a case in point, where consolidated production led to spot price increases exceeding 20% and allocation panic, crippling manufacturers without diversified sourcing.
Concurrently, the premium supplement market is moving toward clinically-backed, condition-specific formulations. The global cognitive health ingredient market, for instance, is projected to grow at a CAGR of 8.5% through 2030. In this landscape, a “high-potency B1” claim using a standard form is merely a table stake, not a differentiator. The fundamental shift required is this: your thiamine selection must be a first-order product strategy, dictated by target health outcomes and brand positioning, not just a procurement spreadsheet.
The Strategic Thiamine Portfolio: A Functional and Commercial Analysis
Each thiamine variant possesses distinct biochemical and commercial profiles. The following analysis moves beyond basic chemistry to clarify the strategic business application of each.
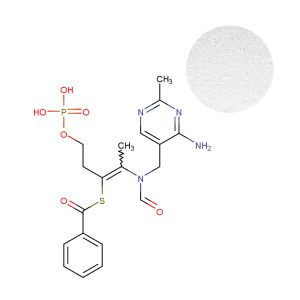
Benfotiamine: Engineering Premium Efficacy for Targeted Health Claims
Benfotiamine is a fat-soluble prodrug designed for superior bioavailability. Its commercial value lies not in being “another B1,” but in its validated mechanism for addressing metabolic stress and neurological health. With a cellular uptake estimated to be 3-5 times higher than water-soluble forms, it delivers the tissue concentrations necessary for therapeutic-level effects.
The pivotal 2024 initiation of the “Benfoteam” Phase II clinical trial in early Alzheimer’s patients is not just a research milestone; it is a market signal. It provides the credible, high-caliber scientific anchoring that premium brands require to justify premium pricing and build defensible marketing narratives. For a product targeting healthy glucose metabolism or peripheral nerve comfort, Benfotiamine allows you to speak in terms of specific biochemical pathways (like transketolase activation and reduction of advanced glycation end-products), rather than generic nutrient support.
Strategic Application & Formulation Insight:
Deploying Benfotiamine Powder effectively requires a synergistic formulation philosophy. For a cognitive health product, pairing it with Citicoline and Phosphatidylserine can create a complementary mechanism for neuronal membrane and neurotransmitter support. For metabolic health, combining it with Alpha-Lipoic Acid targets multiple points in the mitochondrial energy and antioxidant network. The goal is to create an efficacy matrix, where Benfotiamine acts as the cornerstone bioactive, elevating the entire formula’s value proposition. A brand using this approach for a European market entry is better positioned to compile a robust scientific dossier for EFSA health claim consideration.
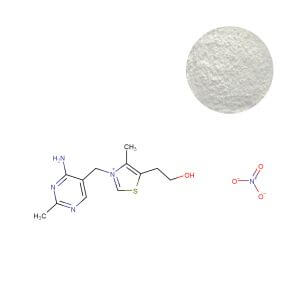
Thiamine Mononitrate: Securing the Supply Chain of a Critical Fortificant
Thiamine Mononitrate’s unmatched stability makes it the irreplaceable choice for mass fortification in foods, beverages, and standard supplement formats. However, its 2024-25 story is a textbook study in concentrated supply chain risk. Industry intelligence indicates that a single geographic region influences the majority of global capacity, turning routine facility maintenance into global market events.
This reality demands a procurement strategy centered on business continuity assurance. The real cost of Mononitrate must be calculated as Total Cost of Ownership (TCO), which includes:
- Disruption Cost: The revenue lost from a production line idled by ingredient shortage.
- Expedite Cost: The exorbitant premiums for air freight during a crisis.
- Quality Risk: The potential compromise of vetting new, unqualified suppliers under pressure.
Strategic Sourcing & Vendor Qualification:
When Thiamine Mononitrate is the technically correct form, your sourcing process must be forensic. Beyond GMP certification, demand transparent answers to:
- Production Geography: “Can you provide audit reports for at least two discrete manufacturing sites?”
- Inventory Policy: “What is your safety stock level for this ingredient, and what triggers your allocation protocol?”
- Market Intelligence: “What is your forecast for lead times over the next 12 months, and what are your key risk indicators?”
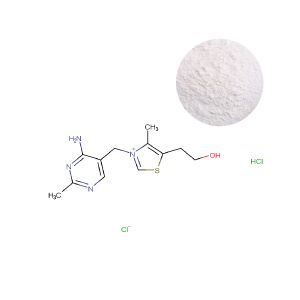
Thiamine Hydrochloride: The Compliance-Driven Choice for Precision Delivery
Thiamine HCl’s primary advantage is its exceptional solubility, making it mandatory for applications where rapid dissolution and absorption are part of the core value proposition. Its strategic importance, however, is amplified in regulated and clinical channels.
For medical nutrition products, therapeutic foods, or injectable solutions, pharmacopeial standards (USP, Ph. Eur.) and FDA guidelines often explicitly specify Thiamine HCl. In these contexts, using Mononitrate is not an option—it is a regulatory non-compliance. This positions HCl as the ingredient of choice for brands targeting hospital formularies, bariatric surgery clinics, or professional sports nutritionists, where evidence of pharmaceutical-grade sourcing is a prerequisite for entry.
Strategic Implementation & Market Access:
Utilizing Vitamin B1 Thiamine HCl is a declaration of precision. In a ready-to-mix electrolyte powder for athletes, its instant solubility is a tangible, marketable benefit over competitors whose products may clump or dissolve poorly. For a pediatric liquid vitamin, it guarantees homogeneous dosing. The formulation imperative is to manage its hygroscopicity through robust packaging (e.g., desiccant-lined pouches) and to highlight its purity and solubility in your technical marketing materials to downstream B2B clients.
An Operational Blueprint: Integrating Strategy into R&D and Procurement
To operationalize this strategic view, companies must institutionalize cross-departmental workflows that prioritize value and risk management over mere cost.
Stage 1: Claim-Driven Ingredient Selection (Marketing & R&D)
Initiate the NPD process with a “Target Outcome Document.” This document answers: What is the single most important health benefit this product will promise? (e.g., “Supports nervous system comfort in healthy glucose metabolism.”). This outcome automatically preselects the ingredient platform—in this case, Benfotiamine—before any formulation begins.
Stage 2: Value-Based Sourcing Protocol (Procurement)
Replace the standard RFQ with a Supplier Capability Assessment. For Benfotiamine, weight scoring toward technical support and clinical dossier access (50%). For Mononitrate, weight scoring toward supply chain transparency and business continuity planning (50%). For HCl, weight scoring toward regulatory documentation and purity specifications (50%). Cost should never account for more than 40% of the total score.
Stage 3: Lifecycle Stability Partnership (Quality & Supply Chain)
Engage your ingredient partner in co-developing a stability and handling profile. This includes:
- Jointly defining ideal storage conditions (temperature, humidity) for bulk material.
- Conducting stress tests on the finished product to validate shelf-life claims.
- Establishing a shared quality alert system for any batch-to-batch variability.
Conclusion: The Defining Question for Leadership
The vitamin B1 landscape has permanently shifted. The molecule is no longer a commodity, but a versatile toolkit. Market leaders will be those who strategically select Benfotiamine to command premium segments, intelligently source Mononitrate to ensure operational continuity, and precisely apply HCl to access regulated, high-trust channels.
The ultimate question for your executive team is not “Who has the cheapest thiamine this quarter?” It is: “What specific, valuable problem are we solving for our end-user, and which thiamine chemistry is the indispensable key to solving it credibly and reliably?” Your answer determines whether you are manufacturing a cost-driven commodity or building a valued, resilient brand. In this paradigm, your ingredient supplier is not a vendor, but a critical strategic partner, integral to your innovation, risk mitigation, and market ambition.
The path forward requires intentionality, from the molecule up.
Share this article
Found this helpful? Share it with others!
Related Products
Products mentioned in this article
Want to learn more?
Explore our products or contact our team for personalized solutions and expert advice.