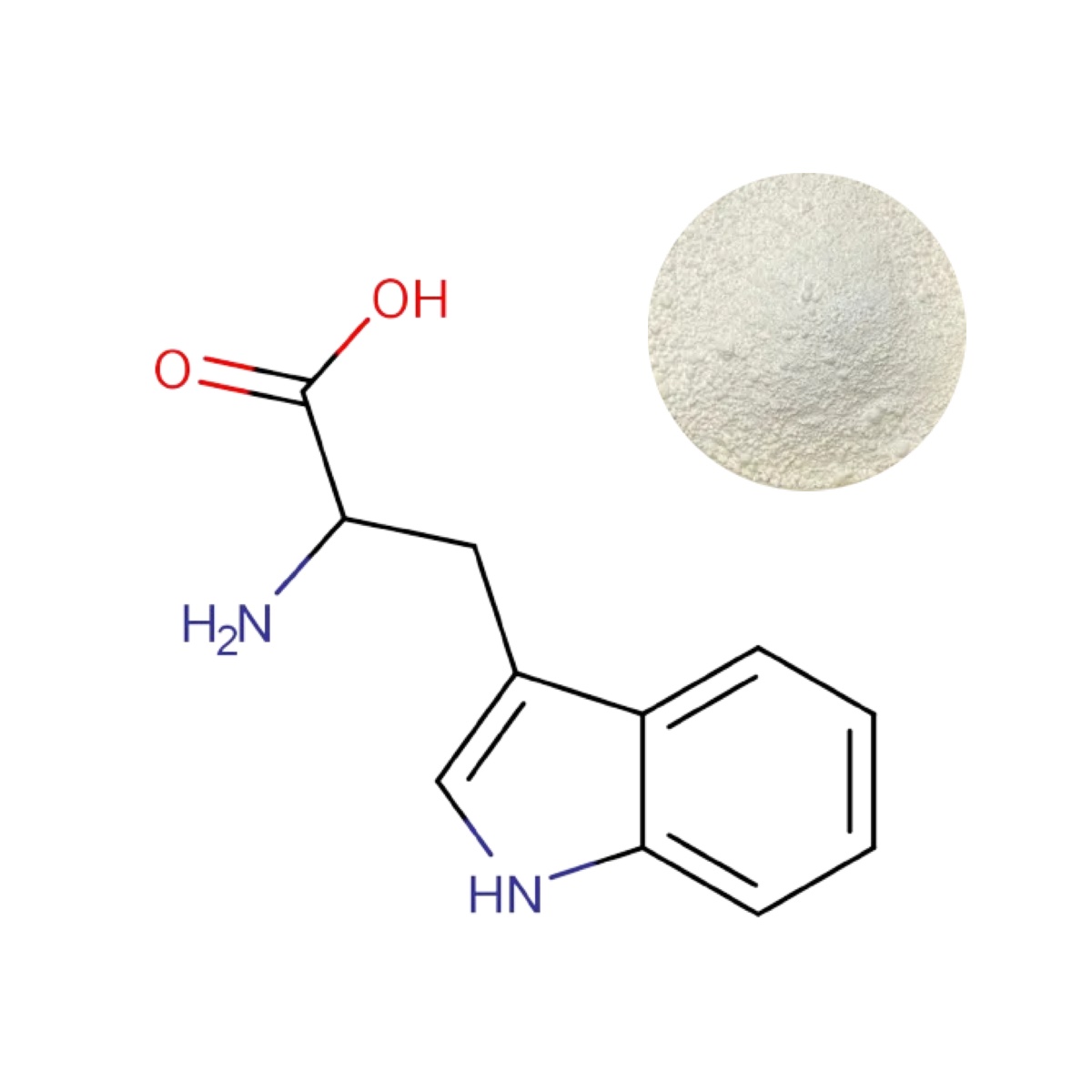
L-Tryptophan Powder
| Product Name | L-Tryptophan Powder |
| CAS Number | 73-22-3 |
| Appearance | White to off-white crystalline powder |
| Purity | 99% min. (by HPLC, on dried basis) |
| Packaging | 1 kg/bag, 5 kg/bag, 25 kg/drum |
| MOQ | 1 kg |
L-Tryptophan Powder: A Formulation Cornerstone for Gut–Brain & Metabolic Health
Formulating differentiated supplements requires moving beyond generic amino acid profiles. Today’s sophisticated brand owners seek ingredients with validated mechanisms—not just historical use—and the supply chain transparency to back every claim. L‑Tryptophan, standardized to ≥99% purity by HPLC, meets this demand. Its role as a precursor to serotonin and melatonin is well established, yet recent clinical insights reveal two compelling new dimensions: gut‑derived hormone modulation and intestinal immune signaling. For R&D and procurement leaders, understanding these mechanisms is the first step toward building product lines that stand apart in crowded categories such as metabolic wellness and gut‑immune support. This article provides a practical framework for integrating this premium ingredient into cutting‑edge formulations while ensuring quality, stability, and regulatory alignment.
Beyond Serotonin: Dual Mechanisms That Redefine L‑Tryptophan’s Value
Most formulators know L‑tryptophan as the substrate for serotonin synthesis. A more complete picture, however, positions it as a strategic lever for two distinct health targets—a distinction that directly impacts how brands position their finished products. Recent human intervention studies have documented its ability to influence both the gut‑brain axis and energy regulation in ways that go far beyond mood support alone.
- Gut hormone activation: A randomized, crossover trial in healthy males showed that duodenal L‑tryptophan administration significantly increased circulating levels of cholecystokinin (CCK), glucagon‑like peptide‑1 (GLP‑1), and peptide YY (PYY) — hormones that collectively promote satiety and reduce subsequent energy intake. Importantly, co‑administration with calcium produced a dose‑dependent amplification of this effect, with 1 g of calcium enhancing energy‑intake suppression by 16.8% compared to tryptophan alone (doi: 10.1016/j.ajcnut.2024.07.006).
- Intestinal immune signaling: A separate controlled study demonstrated that oral L‑tryptophan (3 g/day over three weeks) activates the aryl hydrocarbon receptor (AhR) in the human duodenum—a key regulator of mucosal immunity and barrier function (doi: 10.1152/ajpgi.00306.2023). AhR activation influences local tryptophan metabolism and may underpin gut‑related benefits that are only beginning to be exploited in dietary supplements.
Practically speaking, these findings shift L‑tryptophan from a one‑dimensional sleep and mood ingredient to a versatile building block for products addressing metabolic wellness, stress‑induced eating, and even gut‑immune resilience. For brands, this means the same powder can anchor multiple SKUs—each with a distinct, clinically informed story. This versatility allows R&D teams to reduce ingredient inventories while expanding portfolio reach.
Quality Benchmarks: What Defines a Premium L‑Tryptophan Powder
When sourcing bulk L‑tryptophan, purity alone is insufficient. For procurement teams, the real value lies in consistent physico‑chemical profiles that guarantee predictable performance in finished goods—and that predictability directly translates to lower production variability and fewer batch rejections. A premium ingredient is defined by tight control over a few critical parameters that influence stability, bioavailability, and regulatory acceptance.
| Parameter | Specification | Why It Matters for Formulators |
|---|---|---|
| Assay (on dried basis) | ≥99.0% by HPLC | Ensures batch‑to‑batch consistency and enables accurate dosage calculation. |
| Loss on Drying | ≤0.2% | Ensures optimal flowability for high‑speed encapsulation, directly increasing hourly throughput and lowering the Total Cost of Ownership (TCO) per batch. |
| Residue on Ignition | ≤0.1% | Indicates minimal inorganic impurities; critical for clean‑label formulations. |
| Particle Size | ≥95% through 80 mesh | Affects blend uniformity and dissolution rate—key for tablets and sachets. |
| Heavy Metals | Pb ≤3.0 mg/kg, As ≤1.0 mg/kg, Cd ≤1.0 mg/kg, Hg ≤0.1 mg/kg | Meets global safety limits (USP, EU) without requiring a “pharmaceutical” label. |
From a compliance standpoint, a comprehensive Certificate of Analysis (COA) that includes these metrics—and goes beyond simple purity—is non‑negotiable. It provides the audit trail needed for regulatory filings in the US (GRAS‑listed, NDI‑exempt) and EU (Novel Food compliant). For procurement teams, this level of documentation reduces qualification time and mitigates supply‑chain risk—particularly critical when managing multi‑country distribution.
Formulation in Practice: Stability, Synergy, and Delivery Solutions
Effective use of L‑tryptophan demands attention to its physical and chemical behavior during processing. Its indole ring is susceptible to oxidation, especially under acidic conditions or in the presence of aldehydes. In aqueous systems, pH should be maintained between 5.0 and 8.0 to avoid side reactions. For dry blends, controlling moisture below 5% in the manufacturing environment is a simple yet critical step to preserve assay over the product’s shelf life. For formulators, mastering these details means the difference between a stable, market‑ready product and one that fails stability testing after months of development.
Synergy offers a powerful way to differentiate finished products. Based on the recent calcium‑co‑administration study, a fixed‑dose combination of L‑tryptophan with calcium citrate or malate can be positioned for appetite control and metabolic support—an emerging category that resonates with weight‑management consumers. For brands targeting the weight‑management space, this combination provides a clinically informed mechanism that goes beyond generic “appetite suppressant” claims. To further differentiate, consider the Trp/LNAA ratio: consuming L‑tryptophan away from high‑protein meals maximizes blood‑brain barrier transport, enhancing consumer‑perceived efficacy. The table below outlines two validated combination strategies, each designed to address distinct consumer needs while maximizing the functional synergy of L‑tryptophan:
| Target Benefit | Key Partners | Rationale | Typical Dose Range (per serving) |
|---|---|---|---|
| Sleep quality & relaxation | GABA Powder, L‑Theanine Powder | GABA promotes neural inhibition; L‑theanine enhances alpha brain wave activity. Together with L‑tryptophan’s serotonin precursor role, this triad supports sleep onset and sleep maintenance through complementary pathways. | 100–300 mg L‑tryptophan 50–200 mg GABA 50–200 mg L‑theanine |
| Satiety & metabolic wellness | Vitamin B6 (Pyridoxine HCl), Soluble Fiber (e.g., Inulin, FOS (Fructooligosaccharides)) | Vitamin B6 serves as a critical cofactor in the serotonin synthesis pathway, ensuring efficient conversion of L‑tryptophan. Soluble fiber adds physical bulk, prolongs gastric emptying, and works synergistically with GLP‑1 stimulation to support sustained satiety. | 1–2 g L‑tryptophan 5–10 mg Vitamin B6 2–5 g soluble fiber |
It’s worth noting that while L‑tryptophan’s bitter taste is manageable in capsules or tablets, it can be challenging in ready‑to‑drink beverages. In such cases, microencapsulation or pairing with natural flavors (cocoa, berry) effectively masks bitterness while protecting the molecule from oxidative stress. From a formulation efficiency standpoint, addressing taste and stability early in development reduces costly reformulation cycles later.
Mitigating Risk: The Checklist for Sourcing Bulk L‑Tryptophan
Selecting a supplier for bulk L‑tryptophan is a decision that impacts product lifecycle cost, not just upfront price. An experienced partner should provide more than a COA; they should offer a framework for consistency and traceability. Accessing a full technical dossier within 24 hours—aligned with USP/EP standards—is critical for streamlining internal audits and accelerating your time‑to‑market. For procurement and quality assurance teams, the ability to quickly audit these elements directly affects time‑to‑market and regulatory readiness. Key sourcing considerations include:
- Manufacturing process transparency: Premium material is produced via controlled fermentation, which yields the pure L‑isomer and avoids optical‑resolution impurities associated with chemical synthesis.
- Batch‑specific documentation: Full COAs covering purity, heavy metals, residual solvents, and microbial limits—all performed by accredited methods (USP, AOAC).
- Stability data: Real‑time and accelerated studies that confirm a 24‑month shelf life under recommended storage (cool, dry, away from light).
- Regulatory support: Access to technical dossiers (US FDA GRAS affirmation, EU Novel Food status, allergen statements) that streamline your own product registrations.
From a total‑cost‑of‑ownership perspective, the small premium paid for fully traceable, GMP‑certified material is quickly recovered by avoiding batch rejections, reducing analytical testing overhead, and accelerating time‑to‑market. This is especially true for brands exporting to multiple jurisdictions where documentary evidence is mandatory. An informed sourcing decision therefore becomes a strategic advantage rather than a simple procurement task.
Next Steps for Your Product Development
L‑Tryptophan has evolved from a classic sleep ingredient into a multi‑functional tool that supports gut‑immune health and metabolic balance—two of today’s fastest‑growing supplement categories. By anchoring your formula with a >99% pure, fermentation‑derived powder backed by recent clinical data, you position your brand at the forefront of science‑backed innovation. To de‑risk your development, we offer complimentary samples (10–20 g) along with a complete technical dossier, including batch‑specific COAs and stability summaries. Request your complimentary sample and technical dossier today and move from concept to market with confidence.
Frequently Asked Questions
Share this product
Related Products
Ready to get started?
Contact our team for technical specifications, pricing, and customized solutions.