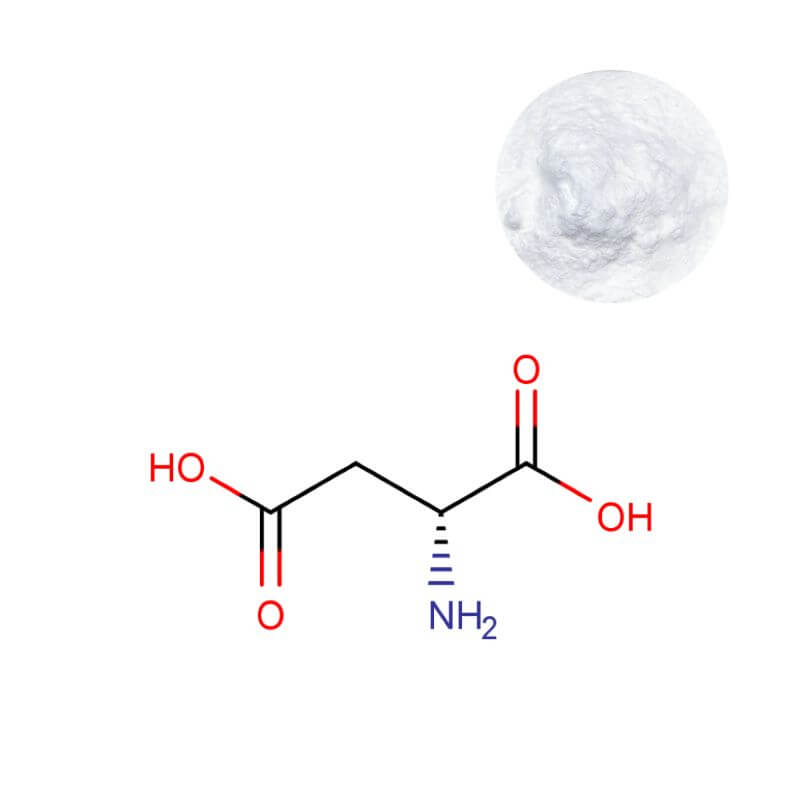
D-Aspartic Acid Powder
| Product Name | D-Aspartic Acid Powder |
| CAS Number | 1783-96-6 |
| Appearance | White crystalline powder |
| Purity | 99.0% min. (by HPLC) |
| Packaging | 1 kg/bag, 5 kg/bag, 25 kg/drum |
| MOQ | 1 kg |
D‑Aspartic Acid Powder: Clinically Validated Support for Testicular & Neuroendocrine Formulations
For brands developing sports nutrition and male health products, the challenge is no longer simply finding a “testosterone booster” ingredient—it is identifying one with a clearly defined mechanism, batch‑to‑batch reliability, and clinical evidence that withstands regulatory scrutiny. Consumer demand for transparency and science‑backed claims has made ingredient selection a strategic decision that directly impacts brand credibility and market positioning. Premium D‑Aspartic Acid (D‑AA) powder, standardized to ≥99% purity with full traceability, addresses this gap. Its role as a signaling molecule in the pituitary and testis is supported by decades of research, while modern analytical methods ensure every shipment meets USP‑NF specifications. This article outlines the science, quality benchmarks, formulation strategies, and sourcing best practices that enable successful product development—translating raw material specifications into commercial advantage.
The Signaling Mechanism: How D‑Aspartic Acid Supports Testicular Activity
Understanding the mechanism behind any functional ingredient is the first step in building a defensible product story. D‑Aspartic Acid accumulates in neuroendocrine tissues, where it acts as a direct regulator of luteinizing hormone (LH) release and Leydig cell steroidogenesis. A 2024 review (doi: 10.3390/cells13161400) details three distinct pathways:
- Central action: D‑AA stimulates LH secretion from the pituitary via cGMP, initiating the hypothalamic‑pituitary‑gonadal axis.
- Peripheral action: It directly activates Leydig cells through cAMP and ERK1/2 signaling, upregulating StAR protein and key steroidogenic enzymes (P450scc, 3β‑HSD, 17β‑HSD).
- Cellular protection: It improves mitochondrial‑associated membrane stability and reduces apoptosis via AMPAR/AKT pathways.
For formulators, this dual mechanism is essential: D‑AA does not simply supply a nutrient—it engages defined hormonal pathways. This molecular clarity allows brands to craft structure/function claims that are defensible and science‑based, distinguishing their products in a crowded category. Moreover, understanding these pathways enables R&D teams to design combination products—pairing D‑AA with mitochondrial cofactors like CoQ10—to leverage additive effects, as demonstrated in recent clinical work. From a commercial standpoint, such mechanistic depth supports premium positioning and helps justify higher price points in a market where consumers increasingly scrutinize ingredient substantiation.
Quality Benchmarks: Beyond Purity to Chiral Integrity & Compliance
For procurement teams evaluating D‑AA powder, a certificate of analysis (COA) must go beyond a simple purity percentage. The real value lies in specifications that directly affect finished product performance and regulatory acceptance. Premium material is defined by:
- Specific rotation – -24.0° to -26.0° (USP <781>). This chiral specification confirms the material is the biologically active D‑enantiomer, not a mixture with inactive L‑AA that would dilute efficacy. For a brand, this means the ingredient delivers the intended clinical effect without requiring higher doses.
- Loss on drying ≤0.5% (USP <731>) – low moisture prevents hydrolysis, directly extending shelf‑life and maintaining potency through the product’s intended lifespan. This translates to fewer consumer complaints and reduced returns.
- Heavy metal limits – Pb, As ≤0.5 mg/kg; Cd ≤0.5 mg/kg; Hg ≤0.1 mg/kg – aligning with global supplement safety standards without relying on pharmaceutical claims.
- Microbiological purity – absence of E. coli, Salmonella, and S. aureus (USP <62>), critical for ready‑to‑mix formats where no terminal sterilization occurs.
- Residual solvents – complying with USP <467> confirms that manufacturing processes (chemical synthesis or fermentation) leave no harmful traces, a key consideration for brands targeting clean‑label claims.
From a compliance standpoint, a COA that includes these parameters effectively pre‑qualifies the ingredient for markets like the US (under DSHEA) and the EU (as a traditional food supplement). For a brand, this reduces the risk of costly reformulation or delayed market entry—a critical advantage when competing in fast‑moving categories.
Formulation in Practice: Dosing, Synergy & Stability Considerations
Translating mechanism into a consumer‑friendly product requires attention to dosage, synergistic partners, and processing conditions. A 2025 randomized controlled trial (doi: 10.4081/aiua.2025.13554) used 2,660 mg/day D‑AA together with ubiquinol (200 mg) and zinc (10 mg) over three months. The study demonstrated significant improvements in total testosterone and progressive sperm motility, providing a validated dosing template for multi‑ingredient formulations.
| Parameter | Practical Insight |
|---|---|
| Typical dosage range | 2.66–3.12 g/day. Lower doses (e.g., 1 g) may be used in combination products; higher doses do not consistently improve outcomes. |
| Synergistic ingredients | Zinc, magnesium citrate, vitamin B6 (as Pyridoxine HCl), and Ubiquinol (CoQ10). Zinc supports steroidogenesis; ubiquinol enhances mitochondrial function in Leydig cells. |
| Processing & stability | Hygroscopic powder—blend in low humidity (≤45% RH). For capsules, use desiccants; for drink powders, the mild acidity is easily masked with natural flavors. |
| Particle size relevance | ≥95% through 80 mesh ensures uniform dispersion in dry blends and accurate dosing in high‑speed encapsulation lines, reducing waste. |
It is worth noting that while some studies in resistance‑trained athletes have shown no acute testosterone elevation, positive results in populations with suboptimal baseline levels highlight the importance of target audience alignment. For brands, this nuance allows precise positioning—for example, focusing on men over 40 or those with fertility concerns—rather than making broad, unsupported performance claims. This targeted approach not only strengthens regulatory defensibility but also improves consumer trust. Additionally, the ingredient’s compatibility with both capsule and powder formats gives formulators flexibility to enter multiple product categories with a single raw material.
R&D Note: D‑AA’s acidic pH (2.5–3.5) requires strategic mineral selection. When formulating with alkaline salts like magnesium carbonate, separate granulation is recommended to prevent premature reactions. Maintaining processing humidity below 45% also prevents micro‑clumping, ensuring fill‑weight precision on high‑speed encapsulation lines.
Mitigating Risk: A Sourcing Checklist for D‑Aspartic Acid Powder
For brands scaling from development to commercial production, a disciplined sourcing approach minimizes downstream surprises. Experienced procurement teams use the following checklist, which shifts the focus from unit price to total cost of ownership:
- Full‑panel COA per batch – includes chiral purity, heavy metals, residual solvents, and full microbiological data, signed by an accredited lab. This eliminates the need for redundant in‑house testing.
- GMP certification – ISO 22000 or FSSC 22000 indicates systematic quality management across production and supply chain, reducing variability that could affect your final product.
- Supply chain transparency – origin of raw materials and production method (chemical synthesis or fermentation) should be documented, with a clear chain of custody. For brands, this enables clean‑label and allergen‑free claims.
- Regulatory documentation – allergen statements, non‑GMO declarations, and stability data (accelerated and real‑time) to support finished product shelf‑life claims. Having these ready cuts months from the development timeline.
- Safety stock & dual sourcing – suppliers that maintain inventory and offer alternative production lines reduce the risk of supply interruptions that could derail launch timelines.
Adopting this checklist transforms ingredient sourcing from a transactional purchase into a strategic partnership. A supplier that proactively provides these materials reduces internal validation time, accelerates launch, and protects brand equity by ensuring raw material consistency—directly contributing to a healthier bottom line. For brands aiming to scale, such diligence becomes a competitive advantage, ensuring that formulation successes can be replicated reliably across production runs.
Strategic procurement should prioritize supply redundancy. A premium partner provides a “Continuity Statement” detailing audited secondary production lines and safety stocks. This transparency mitigates the risk of retail penalties due to stockouts—a value far exceeding marginal unit‑price savings.
Next Steps for Your Product Development
With a clear understanding of D‑Aspartic Acid’s mechanism, quality benchmarks, and formulation best practices, you are positioned to develop a differentiated product. The next step is to evaluate the ingredient firsthand. Request your complimentary 10‑20g sample and comprehensive technical dossier to begin stability studies and formulation trials. Each sample is accompanied by the batch‑specific COA, allowing you to verify every specification outlined above—turning technical data into a launch‑ready formulation with minimized risk.
Frequently Asked Questions
Share this product
Related Products
Ready to get started?
Contact our team for technical specifications, pricing, and customized solutions.