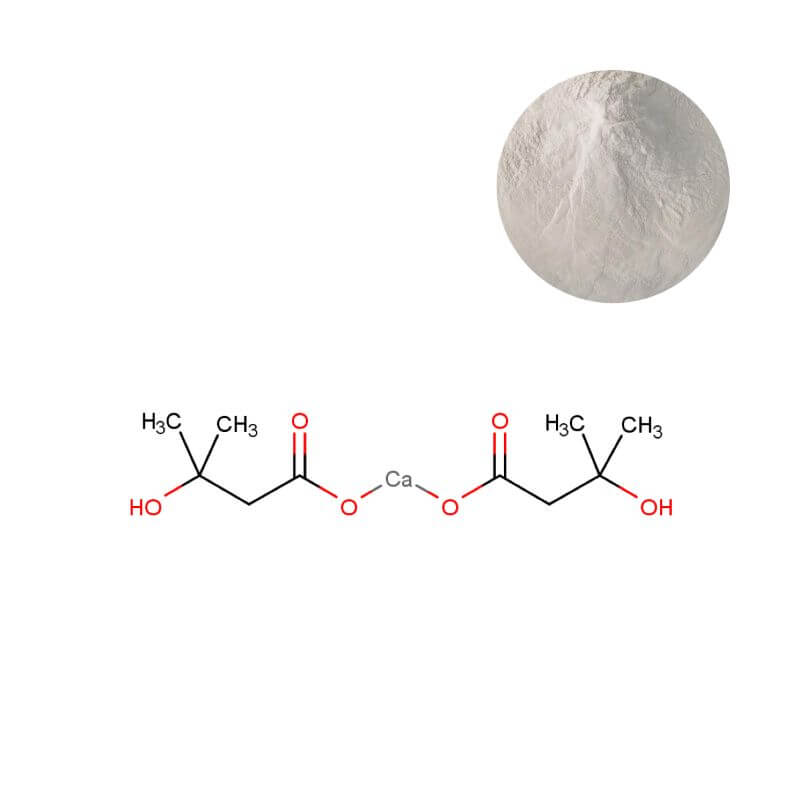
HMB Calcium Powder
| Product Name | HMB Calcium Powder |
| CAS Number | 135236-72-5 |
| Appearance | White or off-white crystalline powder |
| Purity | 99.0% min. (by HPLC) |
| Packaging | 1 kg/bag, 5 kg/bag, 25 kg/drum |
| MOQ | 1 kg |
HMB Calcium Powder: Supporting Muscle Health Formulations for Sports Nutrition and Healthy Aging
For supplement brands aiming to differentiate in the crowded sports nutrition and active aging markets, a generic protein-plus‑vitamin formula no longer delivers a compelling edge. Consumers increasingly seek ingredients with clinically validated mechanisms and clear physiological benefits. β‑Hydroxy‑β‑methylbutyrate (HMB) calcium, a leucine metabolite, has emerged as one such ingredient—backed by decades of research demonstrating its dual role in muscle protein metabolism. Sourced as a high‑purity powder (≥ 99.0 % by HPLC), HMB calcium offers formulators a science‑driven solution for products targeting muscle preservation, recovery, and strength. The following sections translate the technical evidence into actionable sourcing and formulation insights—knowledge that directly impacts a brand’s speed to market, cost structure, and consumer trust.
The Dual Mechanism of HMB: Translating Muscle Science into Tangible Consumer Benefits
HMB acts through two well‑characterised pathways that together create a unique anabolic environment. First, it stimulates muscle protein synthesis by activating the mTOR signalling complex, the same pathway triggered by resistance training and leucine. Second, it attenuates muscle protein breakdown by down‑regulating the ubiquitin‑proteasome system—the primary route for degrading contractile proteins. This dual action distinguishes HMB from single‑pathway ingredients and explains its efficacy across diverse populations.
A comprehensive 2024 position stand from the International Society of Sports Nutrition consolidates the evidence: daily supplementation with 3 g of HMB consistently improves lean mass gains and strength in both trained athletes and untrained individuals initiating exercise (doi: 10.1080/15502783.2024.2434734). For the aging demographic, the same mechanism helps counteract sarcopenia—the gradual loss of muscle mass that begins around age 40. Commercially, this means that an HMB‑based product can support substantiated structure/function claims such as “supports muscle recovery after exercise” and “helps maintain muscle mass during aging,” giving brands a credible platform for consumer communication. In a market where efficacy claims are increasingly scrutinized, having a dual‑mechanism ingredient allows for richer, more differentiated storytelling that resonates with educated buyers.
Beyond Purity: Critical Quality Benchmarks for Sourcing Premium HMB Calcium
While total purity (≥ 99.0 %) is the baseline, professional procurement requires scrutiny of additional specifications that directly affect product performance and safety. A premium‑grade HMB calcium should exhibit:
- Calcium content: 13.5–16.0 % – ensures correct stoichiometry and avoids excess mineral load that could interfere with co‑formulated nutrients.
- Loss on drying ≤ 3.0 % – a stricter limit than the common ≤ 7 %, indicating lower moisture and better flowability. For production, this translates to fewer compression issues in tablet manufacturing, reduced clumping in powder blends, and a longer, more predictable shelf life—directly lowering the risk of batch rejects.
- Heavy metal controls: Pb ≤ 1 mg/kg, inorganic As ≤ 1 mg/kg, Cd ≤ 0.1 mg/kg, Hg ≤ 0.1 mg/kg – meeting these stringent thresholds assures compliance with global regulations (USP, EP, China GB) and protects brand reputation during international market entry.
From a sourcing standpoint, requesting a batch‑specific Certificate of Analysis (COA) that documents these parameters is non‑negotiable. Reliable suppliers provide full disclosure, including residual solvent testing per USP <467>. It is also worth noting that HMB is available in both calcium salt and free acid forms. A 2023 comparative bioavailability study demonstrated that the calcium salt actually exhibits superior bioavailability relative to the free acid, contrary to earlier assumptions (doi: 10.1007/s00726-023-03369-z). For procurement teams, this data removes any perceived advantage of the free acid form, reinforcing the calcium salt as the more commercially reliable and cost‑effective choice for solid‑dosage applications.
Practical Formulation Strategies for HMB Calcium in Supplements
Integrating HMB calcium into finished products requires attention to physical compatibility, taste, and dosage. The ingredient is a white crystalline powder with moderate water solubility (≈ 50 g/L at room temperature) and dissolves more readily in mildly acidic conditions. For dry blends intended for capsules or stick packs, direct incorporation is straightforward. For ready‑to‑drink (RTD) beverages, pre‑dissolving in a portion of the liquid and adjusting pH to 5–6 helps avoid sedimentation.
Key formulation considerations include:
- Typical daily dose: 3 g, based on the majority of clinical trials; the recently updated Chinese regulation (2025) now allows up to 6 g/day, offering flexibility for higher‑strength products targeting advanced users or clinical nutrition applications.
- Synergistic combinations: HMB pairs effectively with Whey Protein Isolate Powder or EAA Powder for enhanced post‑exercise recovery; with Creatine Monohydrate to support both strength and lean mass gains; and with Vitamin D3 and calcium for comprehensive bone‑muscle health in aging formulas—a choice that allows brands to precisely target specific consumer segments, from athletes to active agers, without over‑complicating the formulation.
- Taste masking: HMB calcium carries a characteristic nitrogenous, mildly bitter note. In powder blends and ready‑to‑drink (RTD) formulations, citrus flavour profiles paired with high‑intensity natural sweeteners such as Stevia Extract Powder — or dedicated masking agents — effectively neutralize this off‑note, supporting good consumer acceptability and repeat purchases.
- Stability: The material is non‑hygroscopic when stored properly, but exposure to high humidity during manufacturing should be minimised. In tablet compression, using microcrystalline cellulose or dicalcium phosphate as a filler improves flow and hardness. Validating these process parameters during scale‑up can prevent costly off‑spec batches and preserve the ingredient’s bioavailability.
Practically speaking, running a small‑scale trial blend under controlled humidity (< 45 % RH) and verifying the final product’s dissolution profile ensures that the ingredient’s performance is retained. These steps materially reduce the risk of consumer complaints and protect brand equity.
Ensuring Supply Chain Integrity: Key Considerations for Bulk HMB Calcium Procurement
When buying HMB calcium in bulk, the total cost of ownership extends far beyond the per‑kilogram price. Inconsistent quality—even within a certified specification—can lead to production delays, rejected batches, and damaged consumer trust. Therefore, evaluating potential suppliers against a checklist of operational criteria is a prudent risk‑mitigation strategy.
| Evaluation Criteria | Why It Matters for Your Business |
|---|---|
| Full traceability & batch‑specific COA | Enables rapid response to regulatory inquiries and confirms each lot meets agreed specifications—essential for maintaining certifications and passing customer audits. |
| GMP compliance (e.g., FSSC 22000, ISO 22000) | Indicates a mature quality management system, reducing the likelihood of cross‑contamination or process deviations that could disrupt your production schedule. |
| Capacity and safety stock | A reliable distributor maintains inventory to cover unexpected demand spikes, preventing line stoppages and the associated revenue loss. |
| Documentation support (technical dossier, allergen statement, non‑GMO declaration) | Streamlines your own customer approvals and accelerates time‑to‑market for finished products, giving you a competitive edge. |
A partner that offers these assurances effectively acts as an extension of your quality and regulatory teams. This becomes especially valuable when sourcing for international markets, where compliance with FDA GRAS, EU Novel Food, or China’s new resource food status must be verified. Choosing a supplier with deep regulatory knowledge lowers the barrier to global distribution and allows your team to focus on product innovation rather than firefighting supply issues.
Next Steps for Your Product Development Program
HMB calcium combines a robust scientific foundation with practical formulation versatility—attributes that help brands create credible, high‑performing muscle health products. Whether your target is the elite athlete seeking faster recovery or the active ager aiming to preserve independence, this ingredient delivers on both efficacy and safety. To move from concept to commercial reality, the next step is accessing the technical documentation and material for your own internal evaluation.
We invite qualified professionals to request a complimentary sample and the complete technical dossier, including batch‑specific COA, regulatory status summaries, and formulation guidelines. This package allows your R&D and procurement teams to verify the ingredient’s fit for your product pipeline under real manufacturing conditions—de‑risking the development process and accelerating your path to market.
Frequently Asked Questions
Share this product
Related Products
Ready to get started?
Contact our team for technical specifications, pricing, and customized solutions.